Psoriasis
of interest
are looking at
saved
next event
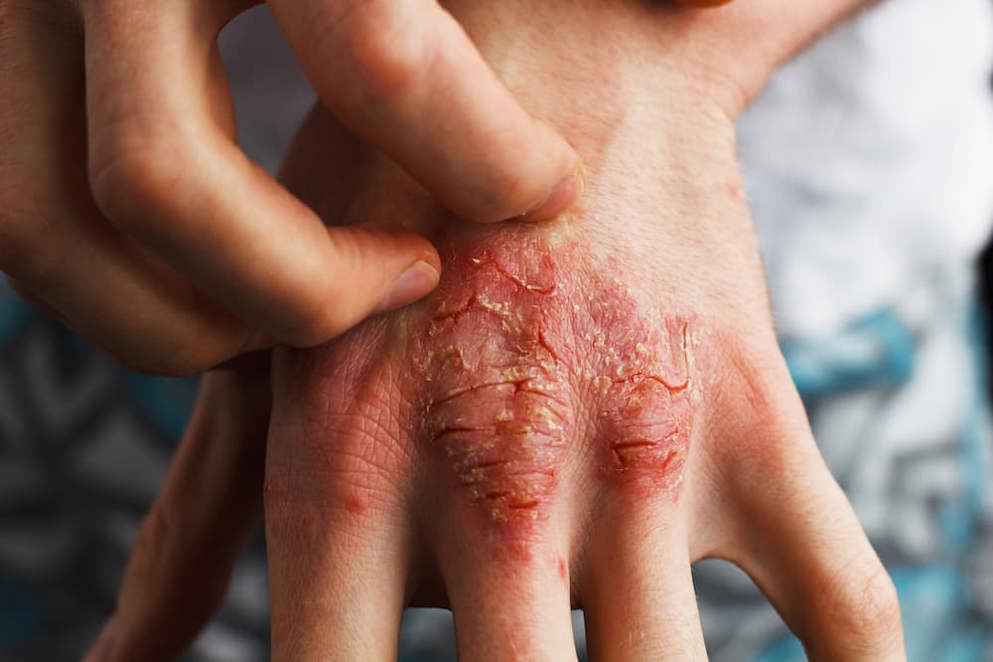
Psoriasis is a common skin condition that affects 1 in 50 people. This chronic inflammatory disease affects men and women equally and can appear at any age.
Childhood psoriasis quiz
What impact does psoriasis have on children’s well-being and how should this condition be managed? Find out with this quiz and learn more about this important topic on Medthority.
Common symptoms include itching and burning sensations, inflamed areas of skin with red patches that may crack and bleed or develop silvery-white scales. In some cases, psoriasis can affect the nails and joints as well as the skin. There are a variety of different forms that the disease can take.
Diagnosis is usually made based on the appearance and distribution of the affected patches of skin. Treatment depends upon individual circumstances. Topical treatment applied to the surface of the skin (topical corticosteroids or vitamin D analogues) is sufficient alone in most patients. For people with more extensive or difficult to treat psoriasis, ultraviolet light treatment (photo/light therapies), oral or injected treatment may be required.
Moderate to severe psoriasis is associated with an increased risk of anxiety and depression as well as a host of comorbidities. It also increases the risk of heart disease and stroke. Furthermore, it can be associated with diabetes, obesity, venous thromboembolism, high cholesterol and high blood pressure as well as inflammatory bowel disease. There is also a small increased risk of skin cancer.
To find out more, navigate through our Learning Zone where we have further information on different forms of psoriasis including inverse psoriasis, pustular psoriasis and guttate psoriasis.

