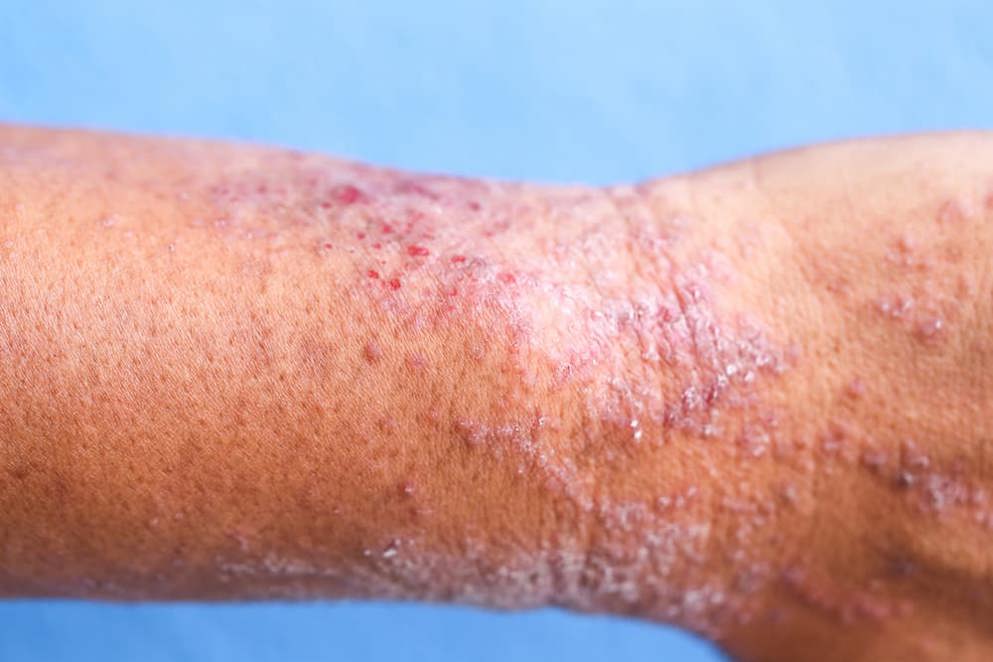
FDA approves Eucrisa (crisaborole) ointment 2% for the treatment of mild to moderate atopic dermatitis.- Pfizer
Pfizer Inc.has announced that the FDA approved Eucrisa (crisaborole) ointment 2%, a novel non-steroidal topical phosphodieterase-4 (PDE-4) inhibitor for the treatment of mild to moderate atopic dermatitis in patients two years of age and older.
Atopic dermatitis (AD) , often called eczema, is a chronic condition impacting nearly 18 million children and adults in the United States. Approximately 90 percent of people living with AD have the mild to moderate form of the condition. Eucrisa is the first and only non-steroidal topical monotherapy that inhibits the PDE-4 enzyme in the skin. Overactive PDE-4 has been shown to contribute to the signs and symptoms of AD. The specific mechanism of action of crisaborole in AD is not well defined.
Comment: Sanofi and Regeneron Pharma are waiting FDA approval of Dupixent (duplimumab) with a PDUFA date in March 2017. Eucrisa is due to launch in January 2017 from Pfizer with a new dedicated dermatology sales force and is likely to be cheaper than Dupixent as it is a topical cream and not a biologic.