Treatment
Depending on the strategy of volume resuscitation, it is often further diluted. Fibrinogen is also reduced (hypofibrinogenaemia), not detectable (afibrinogenaemia) and/or non-functional (dysfibrinogenaemia) in patients with congenital fibrinogen deficiencies. Since fibrinogen deficiencies are known to be associated with an increased risk of excessive bleeding and mortality, treatment involves fibrinogen replacement. Fibrinogen replacement therapies include fresh frozen plasma (FFP), cryoprecipitate and fibrinogen concentrate (FCH), which has been shown to be at least as effective as cryoprecipitate.
Fibrinogen concentrate is at least as effective as cryoprecipitate for treating fibrinogen deficiency; however, practical and safety differences exist.
Here, we highlight clinical trial data on fibrinogen replacement therapies for different clinical settings and introduce practical considerations comparing safety, storage and speed of delivery.
Fresh frozen plasma
Fresh frozen plasma (FFP) is the most commonly used source of coagulation factor replenishment. FFP is prepared by centrifugation of carefully obtained whole blood and contains fibrinogen at a variable concentration of 0.6 g/300mL unit or 2.0 g/L (range = 0.9 to 3.2 g/L), as well as albumin, protein C, protein S antithrombin and tissue factor pathway inhibitor (Theusinger et al., 2011; Kelley & Guzman, 2018). It is stored by freezing to less than -25°C within 8 hours of collection (Stanworth & Tinmouth, 2009).
FFP can be used to treat fibrinogen deficiencies; however, it has several limitations including a low fibrinogen concentration (Theusinger et al., 2011). Large volumes would therefore need to be administered in the case of severe hypofibrinogenemia, increasing the risk of transfusion related complications such as TRALI (transfusion-related acute lung injury) (Benson et al., 2009). It is therefore not recommended as a treatment option for fibrinogen replenishment and should only be used in the absence of cryoprecipitate or fibrinogen concentrate (Franchini & Lippi, 2012; McDonnell, 2018; Spahn et al., 2019).
of interest
are looking at
saved
next event
Cryoprecipitate
Cryoprecipitate is a standard treatment in many countries to replenish fibrinogen in cases of acquired fibrinogen deficiency experienced during trauma involving excessive bleeding, PPH and surgery (Nascimento et al., 2014). Like fresh frozen plasma (FFP), standard preparation of cryoprecipitate does not involve pathogen inactivation. Whilst cryoprecipitate is a standard treatment in the UK, USA, Australia and Canada, most European countries have withdrawn cryoprecipitate as a standard treatment due to the risk of transmitting pathogens and the availability of fibrinogen concentrate as an effective and pathogen-free alternative (Sørensen & Bevan, 2010; Nascimento et al., 2014).
Cryoprecipitate is prepared by the controlled thawing of pooled and frozen FFP at a temperature between 1 and 6 °C (Nascimento et al., 2014). In this process, higher molecular weight proteins precipitate and the remaining soluble proteins are removed as supernatant following centrifugation. The precipitate (cryoprecipitate) is resuspended in a small amount (10 to 20 mL) of remaining supernatant to form a concentrated mixture of fibrinogen, von Willebrand factor, factor VIII, factor XIII and fibronectin (Sørensen & Bevan, 2010). The UK guidelines specify that a minimum of 140 mg of fibrinogen is present in each unit of cryoprecipitate (NICE guideline NG24, 2015), however variations in unit volume exist due to differences in donor fibrinogen levels and cryoprecipitate preparation (Nascimento et al., 2014). Adults are commonly given 200 mL of cryoprecipitate which equates to 2 pools of 5 units each, with an average fibrinogen concentration of 15–17 g/L (Wong & Curry, 2018). This is enough to raise plasma fibrinogen levels by approximately 1 g/L.
Learn more on how cryoprecipitate compares to fibrinogen concentrate for treating fibrinogen deficiency and the associated practical considerations for both treatments in the next sections.
of interest
are looking at
saved
next event
Fibrinogen concentrate (FCH)
Fibrinogen concentrate is a single-factor treatment purified from pools of human plasma and processed further into a powder that allows for easy storage and reconstitution at administration. It is approved for the treatment and prophylaxis of congenital fibrinogen deficiencies such as afibrinogenaemia and hypofibrinogenaemia in many countries including the UK, USA, Canada, Australia and many other European countries (Costa-Filho et al., 2016). It is also approved in Brazil, Uruguay, Taiwan and some European countries such as Germany and Austria as the standard treatment for acquired fibrinogen deficiency experienced during trauma, surgery and postpartum haemorrhage (PPH) (Costa-Filho et al., 2016).
Human fibrinogen concentrate (FCH) is obtained through Cohn fractionation of pooled human plasma. The resulting fraction has high purity and allows for accurate dosing since a defined dose is filled per vial. Reconstitution with sterile water to the desired final concentration for infusion is fast and allows for lower infusion volumes compared to FFP and cryoprecipitate. This latter characteristic reduces the risk of complications related to high transfusion volumes. An additional routine step in the manufacture of fibrinogen concentrate is viral inactivation using techniques such as heat treatment, pasteurisation, nanofiltration, treatment with solvent detergents or a combination of these techniques. This ensures that the risk of viral contamination during treatment remains minimal. The half-life of fibrinogen concentrate was determined as approximately 77 hours in patients of congenital fibrinogen deficiency (Idris et al., 2014). A requirement for repeated doses therefore indicates high levels of fibrinogen consumption or limited production.
There has been recent interest in better understanding the usefulness for fibrinogen concentrate, both for the prevention and treatment of severe bleeding episodes. Also, more comprehensive studies to compare fibrinogen concentrate against the standard cryoprecipitate treatment are underway.
FCH for treatment of congenital deficiency
FCH is the approved standard treatment for acute bleeds and as a prophylactic treatment for congenital fibrinogen deficiencies in the UK, USA, Canada, Australia and many European countries (Costa-Filho et al., 2016). This is because FCH is proven to be safe due to viral inactivation and due to the lower volumes required for infusion (Casini et al., 2016). FCH has been shown to be effective and well tolerated for patients of afibrinogenaemia, hypofibrinogenaemia and dysfibrinogenaemia (Kreuz et al., 2005; Manco-Johnson et al., 2009; Peyvandi, 2009; Casini et al., 2016). FCH has also been shown to improve outcomes when used as a prophylactic treatment prior to surgery for congenital fibrinogen deficiency (Bornikova et al., 2011). This benefit needs to be balanced with the risk of thrombolytic complications and supplementation and is therefore restricted to scenarios where bleeding is expected.
FCH for treatment of acquired fibrinogen deficiency
Fibrinogen concentrate is approved for acquired fibrinogen deficiency in Brazil, Uruguay, Taiwan and some European countries such as Germany and Austria (Costa-Filho et al., 2016). Various studies indicate that fibrinogen concentrate may be useful for treating patients with acquired fibrinogen deficiencies experienced during cardiac and other surgeries, postpartum haemorrhage (PPH), trauma, as well as during liver transplantation and more specifically, for dilutional and consumptive coagulopathies.
of interest
are looking at
saved
next event
FCH in cardiac patients
Low plasma fibrinogen is a predictor for excessive bleeding and mortality risk in cardiac surgery patients (Kozek-Langenecker et al., 2017). Whilst the European Society of Anaesthesiology (ESA) guidelines on management of severe perioperative bleeding recommend a trigger level for fibrinogen supplementation of <1.5–2.0 g/L, there is no consensus on the trigger value specifically in a cardiac setting (Kozek-Langnecker et al., 2017). Studies on the management of perioperative bleeding identified a possible trigger value of 2.15 g/L (2–2.2 g/L) fibrinogen for patients experiencing a severe bleed versus 1.15 g/L as a predictive value for developing a severe bleed (Karkouti et al., 2013; Kindo et al., 2014; Ranucci et al., 2016). Target values in a perioperative setting have not been formally assessed, however studies have aimed to achieve ROTEM-FIBTEM values of 22 mm that corresponds to 3.75 g/L (Rahe-Meyer et al., 2009; Ranucci et al., 2011; Solomon et al., 2011; Rahe-Meyer et al., 2013a). These studies were double-blind randomised trials that assumed either that 4 g fibrinogen should increase plasma concentrations by 1 g/L, or that 1 g of fibrinogen should increase FIBTEM by 2 mm in an average weight person. Ranucci and colleagues identified target values of 2.8 g/L for non-bleeding patients and 3.75 g/L for patients with severe bleeds (Ranucci et al., 2016). They further confirmed that the expected dose to raise fibrinogen levels from 1.15 to 2.80 g/L or from 2.15 to 3.75 g/L is 6.8 g of fibrinogen concentrate or 22 units of cryoprecipitate.
The ESA guidelines on management of severe perioperative bleeding further recommend fibrinogen concentrate (FCH) infusion guided by viscoelastic haemostatic assay monitoring to reduce perioperative blood loss (Kozek-Langnecker et al., 2017). The recommendation is based mainly on the clinical trial and study data, outlined below, which indicate that FCH may reduce the need for allogeneic blood transfusion and is generally safe in a cardiac surgery setting.
A retrospective study to assess fibrinogen recovery parameters after administration of FCH to 39 patients with diffuse bleeding after weaning from cardiopulmonary bypass (CPB) during cardiac surgery found that FCH (mean dose = 6.5 g) increased plasma fibrinogen concentration to more than baseline levels (3.3 g/L, maximum clot firmness [MCF] of 15.5 mm), both on the day of infusion (1.9 to 3.6 g/L, MCF from 10.1 to 20.7 mm) and the following day (1.9 to 4.5 g/L, MCF from 10.1 to 22.3 mm) (Figure 13) (Solomon et al., 2010). Furthermore 90% of patients received no intraoperative transfusion of allogeneic blood products after administration of FCH, suggesting that FCH contributed to the correction of bleeding.
Figure 13. Increase in mean fibrinogen levels in patients (n = 39) after fibrinogen concentrate (FCH) infusion following weaning from cardiopulmonary bypass (CPB) (adapted from Solomon et al., 2010).
In a prospective study of patients with decreased preoperative platelet function undergoing coronary artery bypass graft (CABG), patients with bleeding received either FCH (n = 10) or allogeneic blood products (ABPs) (n = 19) to examine if FCH could help achieve haemostasis when platelet function is low (Solomon et al., 2012). FCH increased FIBTEM MCF (from 10.5 mm to 20.5 mm) and resulted in significantly reduced total transfusion requirement compared to haemostatic treatment with ABPs, indicating that fibrinogen supplementation helps to achieve haemostasis even when platelet function is low (Figure 14). No FCH-related complications were observed and postoperative outcomes were comparable for FCH and ABP treated patients.
Figure 14. Total transfusion units given to coronary artery bypass graft patients with decreased preoperative platelet function receiving either fibrinogen concentrate (FCH, n = 10) or allogeneic blood products (ABP, n = 19) (adapted from Solomon et al., 2012).
A randomised, single-centre, prospective, double-blinded, placebo-controlled clinical trial that investigated the intraoperative use of FCH in 59 patients undergoing complex cardiac surgery found that FCH may be sufficient to control perioperative bleeding in a cardiac surgery setting (Ranucci et al., 2015). They found that patients treated with FCH had a lower rate of ABP transfusion (odds ratio, 0.40; 95% confidence interval [CI], 0.19–0.84, P=0.015) and less postoperative bleeding (300 mL versus 355 mL, P=0.042), compared with placebo (Figure 15) (Ranucci et al., 2015).
Figure 15. (a) Percentage of patients treated with either fibrinogen concentrate (FCH) or placebo that avoided allogeneic blood product (ABP) transfusion and (b) median postoperative bleeding in patients treated with either fibrinogen concentration (FCH) or placebo (adapted from Ranucci et al., 2015).
A single-centre Phase II clinical trial (NCT00701142) including 61 patients undergoing aortic replacement surgery treated patients with either FCH (n = 29) or placebo (n = 32) (Rahe-Meyer et al., 2013b). Avoidance of ABP transfusion was achieved in 13 of 29 (45%) patients in the FCH group, versus 0 of 32 patients in the placebo group (P<0.001), with fewer units needed in the FCH group (median 2 versus 13, P<0.001) (Figure 16). There was no observed safety concern in the FCH group during aortic surgery and the number of intensive care unit (ICU) and hospital-free days were comparable for the two study groups.
Figure 16. Units of allogeneic blood product (ABPs) given to aortic surgery patients who were treated with fibrinogen concentrate (FCH) versus placebo (adapted from Rahe-Meyer et al., 2013b).
The findings of 2013 Rahe-Meyer study were not replicated in a larger Phase III randomised, multi-centre, clinical trial, REPLACE (NCT01475669) that looked at 152 patients undergoing complex cardiovascular surgery (Rahe-Meyer et al., 2013b; Rahe-Meyer et al., 2016). The administration of FCH (n = 78) was associated with an increased need for ABP transfusion compared to placebo (n = 74) (5 units versus 3 units, P=0.026) (Figure 17a). This was in line with a lower avoidance of ABPs for FCH treatment compared to placebo (15.4% versus 28.4%, P=0.047) (Figure 17b). This outcome was despite patients who were treated with fibrinogen concentrate achieving the target FIBTEM MCF of 22 mm compared to no increase for placebo patients.
Figure 17. REPLACE study results. (a) Median units of allogeneic blood products (ABP) supplied to patients treated with either fibrinogen concentrate (FCH) (n = 78) versus placebo (n = 74). (b) Percentage of FCH or placebo treated patients that avoided ABP transfusion (adapted from Rahe-Meyer et al., 2016).
The impact of FCH treatment in newborns, neonates and young infants was also assessed in a recent study on 161 patients (median age 214 days) (Tirotta et al., 2019). In this patient group, FCH administration after cardiopulmonary bypass termination significantly improved clot firmness, as measured by FIBTEM MCF (by 2.9 mm; P<0.001) (Tirotta et al., 2019).
In a meta-analysis of eight randomised clinical trials that included the REPLACE study, FCH supplementation was shown to significantly reduce ABP transfusion versus comparators (risk ratio, 0.64; 95% confidence interval [CI], 0.49–0.83; P=0.001), though no difference in risk mortality was identified (Li et al., 2018). This places doubt over the ABP transfusion result of the REPLACE study and possible limitations to the REPLACE study were identified. These include the clinical practice variance between centres and the use of 5-minute bleeding mass, rather than fibrinogen concentration to guide FCH administration. Furthermore, adherence to the transfusion algorithm was low, at 68% compared to 87% in their earlier Phase II clinical trial and similarly, 62% of patients did not fulfil the bleeding criteria, compared to 19% in the Phase II clinical trial (Rahe-Meyer et al., 2013b; Rahe-Meyer et al., 2016). These limitations were addressed in a post-hoc analysis of the Phase III REPLACE trial, in which patients were stratified according to protocol adherence, pre-treatment fibrinogen levels and whether the patients were among the first three treated at their centre (Rahe-Meyer et al., 2019). The post-hoc analyses revealed that FCH was associated with a trend towards reduced ABP transfusion in treatment-adherent patients who were not among the first three patients to be treated at their centres and with pre-treatment fibrinogen levels of less than 2 g/L.
In an earlier, large, non-randomised, retrospective study on 1,075 patients with bleeding during complex cardiac surgery, data for FCH (264 patients) was compared to placebo (811 patients) (Bilecen et al., 2013). Intraoperative infusion of FCH did not significantly reduce blood loss or ABP transfusion in the intensive care unit (ICU) setting. It is important to note that the patients with a longer cardiopulmonary bypass time or more blood loss were more likely to receive FCH treatment at a low median FCH dose of 2 g, given as a relatively late intervention which may explain the attenuated haemostatic effect. A prospective, randomised trial is needed to better understand outcome. In a later trial study on 120 high-risk cardiac surgery patients with intraoperative bleeding, Bilecen and colleagues randomised patients to receive either FCH (n = 60) or placebo (n = 60), targeted to achieve a plasma fibrinogen level of 2.5 g/L (Bilecen et al., 2017). Whilst 24–hour blood loss was significantly lower for FCH compared to placebo, FCH did not significantly reduce the already modest intraoperative blood loss (50 mL versus 70 mL, P=0.19), with only 2 of 58 FCH-treated patients receiving transfusion compared to 4/59 patients treated with placebo. Unlike their earlier 2013 study, Bilecen and colleagues identified an increase in adverse events experienced by patients treated with FCH, including 2 deaths versus 0 in the placebo group (Bilecen et al., 2017).
There have also been many meta-analysis publications looking at the impact of fibrinogen concentrate on transfusion requirements and mortality in cardiac surgery patients, often including smaller, less rigorous studies. An early meta-analysis study looking at six small clinical trials for cardiac surgery found that there was no statistical impact of FCH treatment on mortality (2.6% vs 9.5%; risk ratio, 0.28; 95% CI 0.03 to 2.33); however, FCH reduced the need for further blood transfusion (risk ratio, 0.47; 95% CI 0.31 to 0.72) (Wikkelsø et al., 2013). A key conclusion from this meta-analysis was that the studies were small and of poor quality. A follow-on systemic review of seven randomised controlled trials (total of n = 268 patients), five of which involved cardiac surgery for both adult and paediatric cases, also reported a similar outcome for fibrinogen concentrate treatment (Lunde et al., 2014). Only two trials from this systemic review reported a significant reduction in mortality and five reported a significant reduction in transfusion requirements. A more recent meta-analysis of eight randomised controlled trials for a total of 597 adult cardiac patients found that using fibrinogen concentrate in the clinic did not affect all-cause mortality (risk ratio, 0.41; P=0.15) but reduced the need for allogeneic red blood cell transfusion (risk ratio, 0.64; P=0.001) (Li et al., 2018). Once again, the authors conclude that larger and better designed trials would help to confirm these findings.
There have been far fewer studies comparing fibrinogen concentrate to the alternative treatments, fresh frozen plasma (FFP) or cryoprecipitate, for acquired fibrinogen deficiency during cardiac surgery. In a small prospective randomised trial of 39 patients, Lance and colleagues compared FFP to partial replacement of FFP with fibrinogen concentrate and found that the latter increased clot formation (Lance et al., 2012). Another small study of 49 patients comparing FFP to fibrinogen concentrate for controlling perioperative bleeding in elective thoracic aortic aneurysm surgery found that fibrinogen concentrate decreased blood loss by 64% and the number of transfusion units by 58% compared to FFP (Yamamoto et al., 2014). Some studies have also compared FCH to other treatments in children and infants. In a prospective randomised study, Galas and colleagues compared fibrinogen concentrate (n = 30) to cryoprecipitate (n = 33) in children undergoing cardiac surgery if bleeding was associated with fibrinogen levels less than 1 g/L. They found that there was no significant difference between FCH and cryoprecipitate in measurements for 48-hour blood loss (320 mL vs 410 mL, P=0.672), need for ABP transfusion or in the increase in fibrinogen levels following administration (Galas et al., 2014). In a more recent, smaller, prospective randomised study on infants (<12 months) undergoing nonemergent cardiac surgery, patients treated with fibrinogen concentrate (n = 29) received less allogenic blood products (1.79 units less) than those treated with cryoprecipitate (n = 25) (95% CI, 0.64 – 2.93; P=0.003) (Downey et al., 2019). In this study, there was no significant difference in secondary outcomes, including ICU and hospital lengths of stay, and adverse events.
FCH was found to be at least as safe as alternative treatments in a cardiac surgery setting (Galas et al., 2014; Fassl et al., 2015; Ranucci et al., 2015; Fominskiy et al., 2016; Rahe-Meyer et al., 2016; Waldén et al., 2020). A longer-term retrospective analysis of bleeding patients undergoing cardiac surgery found no difference in 30 day or 1-year mortality or adverse outcomes in patients treated with FCH (n = 210) compared to patients treated without FCH (n = 781) (Fass et al., 2015) (Table 2).
Table 2. Risks associated with perioperative administration of fibrinogen concentrate (FCH); multivariate logistic regression. Hazard ratios (HR) for mortality and adverse outcome either 30 days or 1 year after fibrinogen concentrate (FCH) treatment (adapted from Fassl et al., 2015)
Fominskiy and colleagues carried out a meta-analysis of fourteen randomised controlled trials investigating the use of FCH in 1,035 surgical patients who were mostly cardiac surgery patients (Fominskiy et al., 2016). Whilst the studies were individually underpowered, the meta-analysis found no difference in the rates of thromboembolic events (TEE) between FCH and control groups, and a possible reduction in all-cause mortality (0.9% for FCH versus 3.5% for the control group, P=0.02). In a more recent, large, retrospective study on a total of 5,408 patients who underwent cardiac surgery, perioperative treatment with fibrinogen concentrate (n = 564) was not associated with an increased risk of thromboembolic complications or death compared to patients who had not received fibrinogen concentrate (Waldén et al., 2020).
Fibrinogen is not indicated for prophylactic treatment in the surgery setting, and there are few studies that address this possible application. During coronary artery bypass surgery, Karlsson and colleagues found that preoperative administration of FCH (2 g dose) reduced postoperative blood loss by 32% (565 +/- 150 vs. 830 +/- 268 ml/12 h, P=0.010) in a prospective randomised pilot study on a total of 20 patients (Karlsson et al., 2009). In a more recent small randomised clinical trial, prophylactic treatment of patients (n = 18) with FCH prior to coronary artery bypass graft surgery significantly reduced the need for blood transfusion and blood products compared to the control group (n = 18) (Zadeh et al., 2019). This finding was not echoed in a slightly larger prospective, randomised study on prophylactic administration in high-risk cardiac surgery patients (Kwapisz et al., 2020). In this study, the cohort had an unusually low transfusion rate in general and patients pre-treated with FCH had significantly higher fibrinogen levels (2.38 vs 1.83 g/L, P<0.001) and improved FIBTEM MCF (27 vs 23 mm, P=0.022) compared to placebo following surgery (Kwapisz et al., 2020). More studies are needed on the value of FCH as a prophylactic treatment in the cardiac surgery setting.
Altogether, it appears that fibrinogen concentrate is safe in a cardiac surgery setting and may result in a reduced need for transfusion of allogeneic blood products. However, since most studies to date are underpowered, more studies are needed, particularly to establish the effect of FCH on mortality, compared to other treatments. A new Phase III, multi-centre, randomised trial (FIBRES trial) seeking to recruit 1200 cardiac surgery patients is currently underway with the aim of directly comparing fibrinogen concentrate to cryoprecipitate (Karkouti et al., 2018).
FCH in postpartum haemorrhage
Postpartum haemorrhage (PPH) may be complicated by acquired coagulopathy as a result of consumption of clotting factors, haemodilution or increased fibrinolysis (Allard et al., 2014; Collins et al., 2016). Low fibrinogen levels in the early phases of PPH correlates with excessive bleeding and the need for blood transfusion (Chartbit et al., 2007; Gayat et al., 2011; Cortet et al., 2012; Poujade et al., 2012).
The International Panel on PPH recommends coagulation screens (including fibrinogen concentration) as soon as persistent PPH is declared to guide treatment decisions (Abdul-Kahir et al., 2014). They recommend the use of FCH or cryoprecipitate for fibrinogen supplementation with the aim to maintain levels >2.0 g/L (Abdul-Kahir et al., 2014). The European Society of Anaesthesiology (ESA) guidelines on management of severe perioperative bleeding recommend assessing fibrinogen levels in patients with bleeding since a level of <2 g/L may identify those patients at risk of developing severe PPH (Kozek-Langenecker et al., 2017). They also recommend fibrinogen replacement in ongoing PPH with hypofibrinogenaemia; however, recommends against pre-emptive fibrinogen replacement (Kozek-Langenecker et al., 2017). It is still unknown whether fibrinogen levels should be maintained at a level that is normal for pregnancy at term (>4 g/L), above the non-pregnant level (>2 g/L) or at an intermediate level. There is very little variation between studies for the possible trigger value of 2 g/L that is indicative of developing postpartum haemorrhage (PPH) (Charbit et al., 2007; Cortet et al., 2012; Collins et al., 2014; Matsunaga et al., 2016). This is reflected in the ESA guidelines on management of severe perioperative bleeding which state that fibrinogen levels of 1.5–2 g/L or FIBTEM values of 12 mm serve as a trigger value for treatment (Kozek-Langenecker et al., 2017). Currently, there is no agreed fibrinogen target value for PPH. In studies that attempted to identify a suitable target value, administration of 1 g fibrinogen concentrate was shown to increase fibrinogen levels by a modest 0.32–0.4 g/L and no differences in bleeding severity was identified, indicating that higher doses may be necessary for the treatment of PPH (Kikuchi et al., 2013; Makino et al., 2015). Indeed, Seto and colleagues recently created an algorithm that supported the use of higher levels of fibrinogen for PPH treatment (Seto et al., 2017).
Fibrinogen concentrate (FCH) has been shown to increase fibrinogen levels and is effective in managing both dilutional and consumptive coagulopathies experienced during PPH (Ahmed et al., 2012; Kikuchi et al., 2013; Mallaiah et al., 2015; Guasch & Gilsanz, 2016). It has also been shown to reduce the need for additional blood component therapy in some settings for managing PPH (Mallaiah et al., 2015; Sahin & Ozkan, 2019).
In a small single-centre study, Kikuchi and colleagues showed a significant increase in plasma fibrinogen levels following FCH supplementation in 18 women who experienced obstetric haemorrhage with an 88% response rate of ‘good’ to ‘moderate’ (Kikuchi et al., 2013). A larger, retrospective observational study including 128 patients who suffered from severe PPH, of whom 115 were treated with 4–6 g FCH and 119 with an average of 6 units FFP, found that fibrinogen levels increased following treatment (Figure 18) (Guasch & Gilsanz, 2016).
Figure 18. Mean fibrinogen level measurements for women prior to labour (n = 267), post-haemorrhage (n = 266) and after fibrinogen concentrate (FCH) supplementation (n = 281) (adapted from Guasch & Gilsanz., 2016).
Matsunaga and colleagues compared FFP to FFP with added fibrinogen concentrate and found that fibrinogen concentrate supplementation reduced the amount of FFP used without impacting on overall blood loss (Matsunaga et al., 2017). In a prospective study evaluating the treatment of patients with major obstetric haemorrhage (blood loss of more than 1500 mL) and coagulopathy (FIBTEM A5 <12 mm), FCH-containing treatment (n = 51) was compared to a shock-pack treatment (n = 42; four units of red blood cells, four units of FFP and one adult dose of platelets). The FCH-containing treatment (containing 3 g dose of FCH) was associated with a reduced need for transfusion and fewer adverse events (Figure 19) (Mallaiah et al., 2015).
Figure 19. (a) Total number of blood product units administered and (b) percentage of patients who experienced transfusion associated circulatory overload (TACO) following treatment with either a fibrinogen concentrate (FCH)-containing treatment (n = 51) or a shock pack (n = 42) (adapted from Mallaiah et al., 2014).
Earlier studies indicated that the efficacy of fibrinogen concentrate was similar to cryoprecipitate for treating PPH patients. Ahmed and colleagues carried out an observational study on PPH patients receiving fibrinogen replenishment from either FCH (n = 20) or cryoprecipitate (n = 14) (Ahmed et al., 2012). They found that FCH was just as efficacious as cryoprecipitate in correcting hypofibrinogenaemia. Whilst they identified no significant benefit of fibrinogen concentrate over cryoprecipitate, they noted a stronger correlation between fibrinogen levels and dose for FCH (r=0.5) compared to cryoprecipitate (r=0.32).
Whilst FCH is not indicated for patients with normal fibrinogen levels, the FIB-PPH trial was the first prospective study to investigate the value of FCH as a pre-emptive treatment for postpartum haemorrhage (Wikkelsø et al., 2015). The findings from this double-blinded, randomised, placebo-controlled study indicated no advantage in using 2 g FCH (n = 123) over placebo (n = 121) as a pre-emptive treatment, including no difference in mortality rates and no difference in blood transfusion requirements (20% versus 22% in the placebo) despite a significant increase in fibrinogen levels for the FCH group. In this study, a low dose of FCH (2 g) was used irrespective of initial fibrinogen levels and body weight, and patients with massive bleeding were not included in the study due to lack of consent (Wikkelsø et al., 2015). Collins and colleagues aimed instead to treat hypofibrinogenaemia with FCH early, in order to prevent the onset of severe PPH (Collins et al., 2017). In this randomised controlled trial of patients who lost between 1000 to 1500 mL of blood, and who possessed a FIBTEM A5 ≤15 mm, patients were treated with either FCH (n = 28) or placebo (n = 27). FCH administered at a trigger level of FIBTEM A5 ≤15 mm (fibrinogen >2 g/L) led to a significant increase in plasma fibrinogen level, but no improvement in the percentage of patients who needed ABP transfusion, and no difference in the amount of blood lost (Figure 20). In a subgroup analysis of patients with FIBTEM A5 ≤12 mm, there were modest, non-significant improvements in blood loss and transfusion volumes (Collins et al., 2017). Altogether, the data indicate that fibrinogen replacement is not required at FIBTEM A5 >12 mm or fibrinogen >2 g/L.
Figure 20. (a) Fibrinogen levels, (b) percentage of patients who received allogeneic blood product (ABP) transfusion and (c) amount of blood lost for patients treated with fibrinogen concentrate (FCH) or placebo (adapted from Collins et al., 2017).
Several studies have found that FCH demonstrates an acceptable safety profile and shows no increased risk of thromboembolic events compared with haemostatic treatments that do not include FCH (Table 3) (Chauleur et al., 2008; Ahmed et al., 2012; Makino et al., 2015; Wikkelsø et al., 2015; Collins et al., 2017).
Table 3. Percentage of postpartum haemorrhage patients who experienced thromboembolic events following either control or fibrinogen concentrate (FCH) treatment, by indicated study
FCH in trauma
Trauma is a major cause of death and accounts for approximately 10% of all deaths globally each year (World Health Organisation). Approximately one third of bleeding trauma patients display signs of coagulopathy that is known to increase the risk of multiple organ failure and mortality (Frith et al., 2010; Spahn et al., 2019). Trauma-induced coagulopathy may arise from a combination of different factors including shock, tissue damage, thrombin-thrombomodulin-complex generation and the activation of anticoagulation and fibrinolytic pathways (Spahn et al., 2019). Pre-existing factors such as age, genetics, comorbidities and pre-injury medication can further influence the onset of coagulopathy in trauma patients (Spahn et al., 2019).
Low fibrinogen levels in a trauma setting has been associated with an increased risk of mortality and a 1 g/L increase in fibrinogen has been shown to reduce the odds of death by 22% (Rourke et al., 2012). The European Task Force for Advanced Bleeding Care in Trauma Guidelines states that early and repeated monitoring of haemostasis is recommended using traditional laboratory tests (including Clauss fibrinogen level) and/or point-of-care prothrombin time/International normalised ratio and/or viscoelastic methods (Recommendation R25; Spahn et al., 2019).
For the initial treatment of patients with expected massive haemorrhage (initial coagulation resuscitation), the guidelines recommend (Recommendation R24; Spahn et al., 2019):
- Fresh frozen plasma (FFP) or pathogen-inactivated FFP in a FFP-red blood cells (RBC) ratio of ≥1:2, as needed or
- Fibrinogen concentrate (FCH) and RBC
The guidelines recommend fibrinogen supplementation for patients with major bleeding with viscoelastic signs of functional fibrinogen deficit or fibrinogen levels of ≤1.5 g/L. Recommended fibrinogen supplementation (Recommendation R28; Spahn et al., 2019):
- FCH (3–4 g is suggested initially) or
- Cryoprecipitate (equivalent to 15–20 single donor units)
- Repeat doses should be guided by fibrinogen levels, assessed by viscoelastic monitoring and laboratory tests
In a single-centre, randomised, double-blind, parallel-group study, adults with severe trauma who required blood transfusion received either placebo (n = 24) or a 6 g dose of FCH (n = 21) (Nascimento et al., 2016). The results of this feasibility study found that FCH increased plasma fibrinogen levels by 0.93 g/L one hour following transfusion, achieving the normal range (>2 g/L) throughout the resuscitation period. The increase was furthermore significantly different to placebo between 1 to 12 hours following infusion (P<0.01) but not at 24 or 48 hours (Figure 21). At later timepoints beyond 24 hours, the acute phase response, during which the liver upregulates fibrinogen synthesis, is likely to account for the majority of plasma fibrinogen present in trauma patients, even when FCH was administered (n = 242) versus no FCH (n = 193) (Schlimp et al., 2016).
Figure 21. Plasma fibrinogen concentration in severe trauma patients following treatment with either fibrinogen concentrate (FCH) (n = 21) or placebo (n = 24) (adapted from Nascimento et al., 2016).
Various studies have looked at the effect of FCH treatment for trauma patients including multiple organ failure, mortality and the occurrence of adverse events (Table 4). Overall, studies on trauma patients indicate that multiple organ failure is similar or lower with FCH treatment compared to no FCH (Nienaber et al., 2011; Nascimento et al., 2016; Innerhofer et al., 2017; Akbari et al., 2018). However, one study indicated that the occurrence of multiple organ failure was higher in patients treated with FCH (Wafaisade et al., 2013). Mortality was found to be similar to, or lower for FCH-treated trauma patients compared to those treated without FCH (Nienaber et al., 2011; Schöchl et al., 2011; Wafaisade et al., 2013; Nascimento et al., 2016; Innerhofer et al., 2017; Akbari et al., 2018; Curry et al., 2018; Mengoli et al., 2018). Finally, a significantly reduced number of red blood cell (RBC) transfusion units were required for trauma patients treated with FCH-based treatment versus those who had no FCH (Nienaber et al., 2011; Schöchl et al., 2011; Innerhofer et al., 2017; Akbari et al., 2018). However, no significant differences in RBC requirements were observed in some other studies (Wafaisade et al., 2013; Nasciemento et al., 2016; Curry et al., 2018). In one study, Itagaki and colleagues found that early administration of FCH to severe trauma patients (n = 31 matched pairs) had a significantly improved in-hospital survival rate compared to the control group (P=0.013) (Itagaki et al., 2020). However, the FCH group required significantly higher amounts of red blood cells and FFP during the first 6 hours of admission, though transfusion amounts were similar between 6 and 24 hours following admission (Itagaki et al., 2020). The findings of the above-mentioned studies are summarised in Table 4.
Table 4. Study findings on multiple organ failure, mortality, transfusion and adverse events for trauma patients treated with fibrinogen concentrate (FCH)-containing treatment versus those treated without FCH-containing treatment.
Innerhofer and colleagues ran a single-centre, parallel-group, open-label randomised trial (RETIC; NCT01545635) on patients of trauma induced coagulopathy to compare FFP to coagulation factor concentrates (CFC; primarily fibrinogen concentrate, or prothrombin complex concentrate and/or FXIII) (Innerhofer et al., 2017). They found that 52% of FFP patients (n = 44) and 4% of CFC patients (n = 50) (odds ratio, 25.34; 95% CI, 5.47–240.03, P<0.0001) required rescue therapy, the former leading to a termination of the trial for safety reasons (Figure 22). 33% of FFP patients required a massive transfusion compared to 12% in the CFC group (odds ratio, 3.04; 95% CI, 0.95–10.87, P=0.042) (Figure 22). These findings indicate that early or first-line treatment with a combination of multiple factor concentrates including FCH may improve treatment outcome.
Figure 22. Percentage of trauma patients who required (a) rescue therapy or (b) a massive blood transfusion following treatment with fibrinogen concentrate containing coagulation factor concentrates (CFC, n = 50) or fresh frozen plasma (FFP, n = 44) (adapted from Innerhofer et al., 2017).
Overall, fibrinogen concentrate treatment in trauma patients appears to reduce the need for transfusion, lowers the rate of multiple organ failure and either lowers or doesn’t impact on mortality (Table 4). More and larger studies in the trauma setting would be beneficial. Further studies comparing single factor agents to multi-factor concentrate treatments should also be of interest.
FCH in other forms of acquired hypofibrinogenaemia
Most studies that address fibrinogen replenishment in the management of coagulopathy focus on cardiac surgery, postpartum haemorrhage and trauma. There are also a handful of studies that focus on alternative scenarios, including liver transplantation, other types of surgery and within a specific coagulopathy setting; for instance, for dilutional coagulopathy or disseminated intravascular coagulation (DIC).
Dilutional coagulopathy following that administration of fluids is a major challenge for anaesthetists during surgery. It leads to a significant reduction in clotting factors which impairs both fibrinogen polymerisation and clot strength, thereby increasing the risk of postoperative haemorrhage (Fries et al., 2006). In an early in vitro study, Fries and colleagues induced dilutional coagulopathy (60% dilution) in blood taken from 5 healthy volunteers by adding lactated Ringer’s solution, 4% modified gelatin solution, or 6% hydroxyethyl starch (HES) 130/0.4 to measure the effect of FCH addition (Fries et al., 2006). FCH addition decreased clotting time and increased clot firmness and fibrin polymerisation in a dose-dependent manner. Another in vitro study diluted blood from 8 healthy volunteers by 60% using either saline [NaCl 0.9%] or 6% HES 130/0.4 (De Lorenzo et al., 2006). In this study, the addition of FCH reconstituted clot firmness in samples diluted by saline but only led to a minor effect on clot firmness in samples diluted by HES 6%. In a porcine model of dilutional coagulopathy, FCH administered significantly reduced blood loss compared to the animals that were treated with saline (1,100 mL versus 2,010 mL, P=0.018) (Fries et al., 2005). In patients undergoing radical cystectomy who experienced dilutional coagulopathy following infusion of hydroxyethyl starch (HES), FCH was found to significantly improve maximum clot firmness (P<0.05), velocity of clot formation (P<0.05) and significantly reduce the need for post-operative red blood cell (RBC) transfusion (2/10 patients in FCH group vs 8/10 in placebo group; P=0.023) (Fenger-Eriksen et al., 2009).
FCH was also tested on 66 spinal surgery patients who were randomly assigned to receive either modified gelatin solution, HES 130/0.4 or Ringer lactate solution (Mittermayr et al., 2007). FCH was administered when fibrinogen polymerisation was critically decreased, measured by a FIBTEM-MCF <7 mm. A total of 13 patients receiving HES or gelatin (but none in the Ringer lactate group) needed FCH which increased fibrinogen levels to maintain borderline clot firmness, despite ongoing blood loss. In a small study on children (n = 9) experiencing dilutional coagulopathy whilst undergoing craniosynostosis surgery, FCH administration was shown to improve fibrinogen polymerisation and total clot strength (Haas et al., 2008). A later study on children undergoing craniosynostosis or scoliosis surgery additionally found that prophylactic treatment with FCH at a trigger FIBTEM MCF value of <13 mm decreased bleeding and transfusion requirements during craniosynostosis surgery but not scoliosis surgery (Haas et al., 2015). The Dutch Working Group on Craniosynostosis recommends fibrinogen concentrate over FFP and cryoprecipitate for the management of blood loss in craniosynostosis surgery patients characterised by dilutional and consumptive coagulopathies (Mathijssen et al., 2015).
Disseminated intravascular coagulation (DIC) increases the risk of profuse haemorrhaging and arises through the activation of the coagulation cascade and subsequent depletion, or consumption of platelets and clotting factors. DIC has been observed in sepsis, malignancy, trauma, liver disease and vascular anomalies. In an open, placebo-controlled study of a rat sepsis model, DIC was induced by injection of bacterial lipopolysaccharides (LPS) (Kaspereit et al., 2004). FCH administration was able to restore fibrinogen levels in a dose-dependent manner and ameliorate coagulopathy.
The European guidelines for the management of DIC recommend fibrinogen concentrate (3 g) or cryoprecipitate for cases of severe hypofibrinogenaemia (<1 g/L) that persists despite FFP replacement therapy (Levi et al., 2009). The International Society on Thrombosis and Haemostasis (ISTH) guidelines for the management of DIC also recommend fibrinogen concentrate or cryoprecipitate for actively bleeding patients with persisting severe hypofibrinogenaemia (<1.5 g/L) despite FFP replacement (Wada et al., 2013). The ISTH guidelines for the management of cancer-associated DIC recommends cryoprecipiate or fibrinogen concentrate in actively bleeding cases with persistently low fibrinogen levels (<1.5 g/L) despite supportive measures which include platelets, FFP and prothrombin complex concentrate (PCC) as recommended (Thachil et al., 2015). The Italian Society for Haemostasis and Thrombosis (SISET) guidelines also recommend the use of fibrinogen concentrate or cryoprecipitate in cases of severe hypofibrinogenaemia (<1 g/L) that persists despite FFP replacement (Di Nisio et al., 2012).
Few studies exist for other types of surgery. A small prospective, randomised, controlled Phase II study on cytoreductive surgery for pseudomyxoma peritonei found that 4 g fibrinogen concentrate (n = 11) was at least as efficacious as 10 units (2 pools) of cryoprecipitate (n = 13), with similar levels of blood loss (Roy et al., 2018). In a small randomised study of 60 men, Soleimani and colleagues found no difference in blood loss for placebo versus a low dose of preoperative fibrinogen concentrate administered to patients undergoing transurethral resection of the prostate (Soleimani et al., 2017). Fibrinogen concentrate was shown to reduce perioperative bleeding in patients undergoing total hip arthroplasty (Najafi et al., 2014). More recently, a prospective, randomised Phase II study (FORMA-05) on patients undergoing cytoreductive surgery for pseudomyxoma peritonei found that fibrinogen concentrate (n = 21) demonstrated noninferior efficacy (P=0.0095) compared to cryoprecipitate (Roy et al., 2020).
FCH supplementation has also been shown to improve fibrinogen plasma levels in patients who suffer from chronic severe hypofibrinogenaemia, such as patients with haemotalogical malignancy or hepatic insufficiency. The European Association for the Study of Liver (EASL) guidelines state that although indications for significant active haemorrhage in the specific setting of acute liver failure are not available, it seems reasonable to target plasma fibrinogen levels 1.5–2 g/L by infusing FCH at an initial dose of 25–50 mg/kg body weight, and a platelet count >60,000/µL (European Association for the Study of the Liver, 2017). In an ex vivo study, FCH was added to plasma taken during orthotopic liver transplantation (OLT) (n = 15) or plasma from healthy controls (n = 15) (Groeneveld et al., 2015). FCH addition to the OLT plasma substantially improved fibrin clot structure. In a retrospective study of 212 patients undergoing OLT, patients who received PCC -/+ FCH versus control had similar blood product requirements, but more platelet transfusion (3.1 versus 2.2, P=0.005) (Colacecchia et al., 2017). This contrasts with another study which found that fibrinogen concentrate reduced the need for blood products (Noval-Padillo et al., 2010). Pre-emptive treatment with FCH also did not influence transfusion requirements during liver transplantation (Sabate et al., 2016).
Patients of paediatric acute lymphoblastic leukaemia (ALL) can experience hypofibrinogenaemia as a complication of asparaginase treatment which leads to the concurrent depletion of fibrinogen and antithrombin, thereby increasing the risk of bleeding and haemorrhagic complications. A small study on 7 children with ALL and acquired severe hypofibrinogenaemia (<0.5 g/L) secondary to asparaginase chemotherapy found a modest increase in fibrinogen levels following FCH administration (0.7 g/L) (Giordano et al., 2014).
Altogether, larger studies with greater statistical power are required to better compare the available treatments for hypofibrinogenaemia in distinct settings discussed.
Practical considerations for fibrinogen replenishment: a comparison
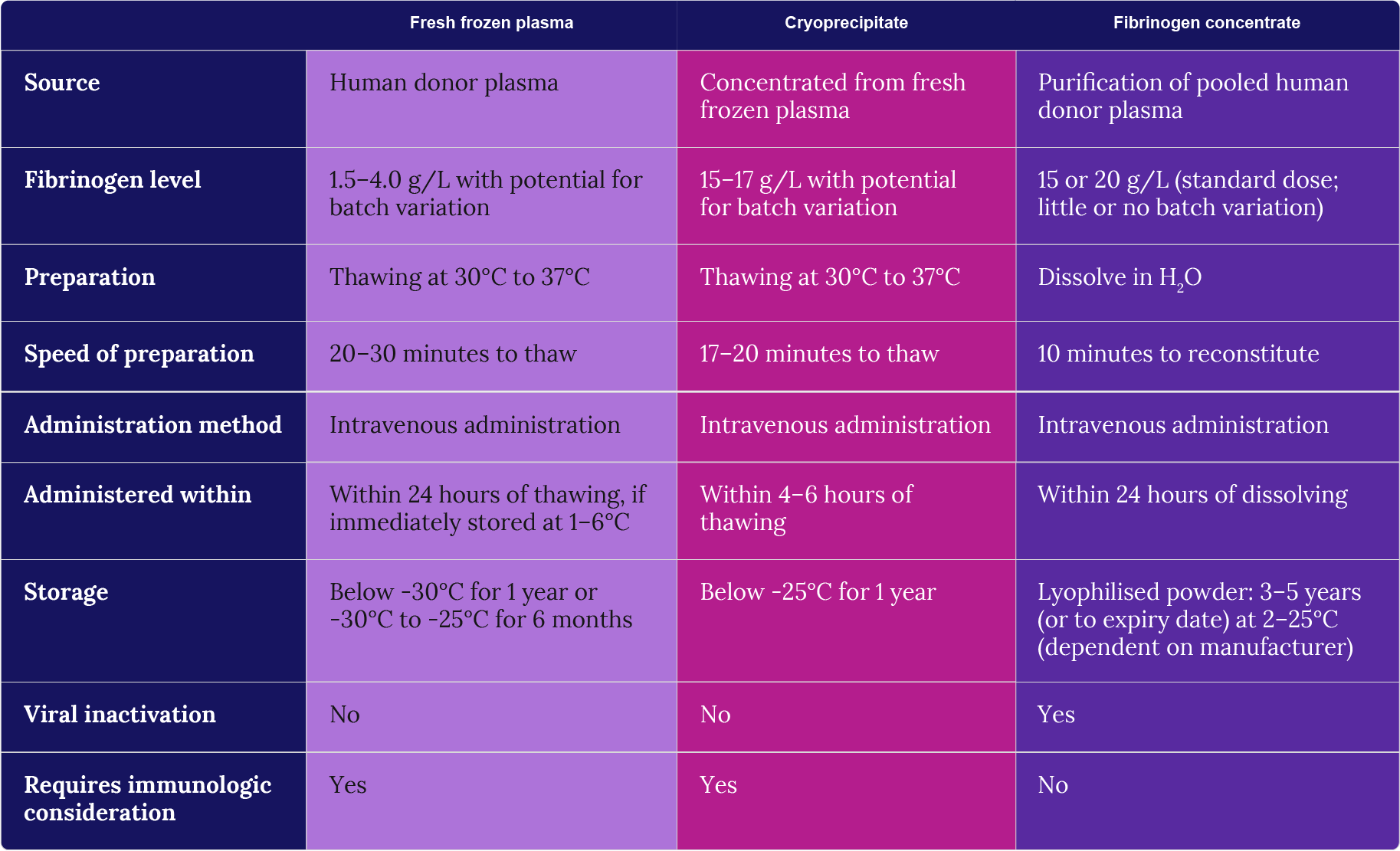
Unlike FFP and cryoprecipitate, fibrinogen concentrate is a single-factor agent. Despite this, fibrinogen concentrate has been shown to be as effective as FFP and cryoprecipitate, indicating that additional coagulation factors present in FFP and cryoprecipitate are unlikely to impact on treatment outcome for most cases. In terms of replenishment, there appears to be no additional clinical benefit of using fibrinogen concentrate over FFP or cryoprecipitate. The major differentiating factors to consider in the clinic is therefore cost, speed of preparation, storage and safety (Table 5).
Table 5: Comparison of fresh frozen plasma (FFP), cryoprecipitate and fibrinogen concentrate (FCH) (adapted from Wong & Curry, 2018).
FFP and cryoprecipitate are more widely available and cheaper to source than fibrinogen concentrate; however, indirect costs for preparation, transportation, storage and associated wastage are often overlooked and need to be considered in evaluating real-term cost-effectiveness (Sørensen & Bevan, 2010; Nascimento et al., 2014; Wong & Curry, 2018). A recent study calculated that after considering 28% wastage for cryoprecipitate, a further 44% reduction in cost of fibrinogen concentrate or a significant reduction in time spent at ICU would be needed to match the cost of cryoprecipitate (Okerberg et al., 2016). More recently, a cost analysis in the Netherlands found that fibrinogen concentrate is a cost-saving option for the management of bleeding during cardiac surgery when compared to fresh frozen plasma (Kelly et al., 2019). While the acquisition cost was higher for fibrinogen concentrate, other hospital-based costs were found to be lower, leading to a 21.1% reduction in overall cost of FCH compared to FFP (Kelly et al., 2019).
Significant benefits in safety and effectiveness are required to make fibrinogen concentrate economically viable (Wong & Curry, 2018). Product preparation and time to administer impact on the safety of use. The amount of fibrinogen in fibrinogen concentrate is standardised whereas the amount of fibrinogen in cryoprecipitate varies on average between 15–17 g/L but has also been shown to vary drastically from 3–30 g/L (Nascimento et al., 2014; Wong & Curry, 2018). Unlike with FFP and cryoprecipitate, the preparation of fibrinogen concentrate involves viral inactivation and removal of antibodies and antigens that can trigger an allergic response. The former means that fibrinogen concentrate carries a significantly reduced risk of viral infection and the latter eliminates the need for blood group matching. In terms of preparation, FFP and cryoprecipitate require blood group matching and approximately 17–20 minutes for thawing at 30–37°C. Fibrinogen concentrate takes approximately 10 minutes to reconstitute and is administered faster and at lower volumes (100 mL) compared to FFP and cryoprecipitate, thereby reducing the risk of transfusion volume-related complications. Reported adverse events are similar (Wong and Curry, 2018). There are clear advantages for using fibrinogen concentrate over standard cryoprecipitate therapy, however, further studies are needed to ascertain true costs, safety and effectiveness of fibrinogen concentrate compared to cryoprecipitate.
of interest
are looking at
saved
next event
Treatment references
Abdul-Kadir R, McLintock C, Ducloy AS, El-Refaey H, England A, Federici AB et al. Evaluation and management of postpartum hemorrhage: consensus from an international expert panel. Transfusion. 2014;54:1756–68.
Ahmed S, Harrity C, Johnson S, Varadkar S, McMorrow S, Fanning R et al. The efficacy of fibrinogen concentrate compared with cryoprecipitate in major obstetric haemorrhage—an observational study. Transfus Med. 2012; 22: 344–9.
Akbari E, Safari S, Hatamabadi H. The effect of fibrinogen concentrate and fresh frozen plasma on the outcome of patients with acute traumatic coagulopathy: A quasi-experimental study. Am J Emerg Med. 2018;36:1947–1950.
Allard S, Green L, Hunt BJ. How we manage the haematological aspects of major obstetric haemorrhage. Br J Haematol. 2014;164:177–88.
Benson AB, Moss M, Silliman CC. Transfusion-related acute lung injury (TRALI): a clinical review with emphasis on the critically ill. British journal of haematology. 2009;147:431–43.
Bilecen S, Peelen LM, Kalkman CJ, Spanjersberg AJ, Moons KG, Nierich AP. Fibrinogen concentrate therapy in complex cardiac surgery. J Cardiothorac Vasc Anesth. 2013;27:12–7.
Bilecen S, de Groot JA, Kalkman CJ, Spanjersberg AJ, Bruinsma BB, Moons KG et al. Effect of fibrinogen concentrate on intraoperative blood loss among patients with intraoperative bleeding during high-risk cardiac surgery: a randomized clinical trial. JAMA. 2017;317:738–47.
Bornikova L, Peyvandi F, Allen G, Bernstein J, Manco-Johnson MJ. Fibrinogen replacement therapy for congenital fibrinogen deficiency. J Thromb Haemost. 2011;9:1687–704.
Casini A, de Moerloose P, Neerman-Arbez M. Clinical features and management of congenital fibrinogen deficiencies. Semin Thromb Hemost. 2016;42:366–74.
Chauleur C, Cochery-Nouvellon E, Mercier E, Aya G, Marès P, Mismetti P et al. Analysis of the venous thromboembolic risk associated with severe postpartum haemorrhage in the NOHA First cohort. Thromb Haemost. 2008;100:773–9.
Charbit B, Mandelbrot L, Samain E, Baron G, Haddaoui B, Keita H et al. The decrease of fibrinogen is an early predictor of the severity of postpartum haemorrhage. J Thromb Haemost. 2007;5:266–73.
Colavecchia AC, Cohen DA, Harris JE, Thomas JM, Lindberg S, Leveque C et al. Impact of intraoperative factor concentrates on blood product transfusions during orthotopic liver transplantation. Transfusion. 2017;57:3026–34.
Collins PW, Solomon C, Sutor K, et al. Theoretical modelling of fibrinogen supplementation with therapeutic plasma, cryoprecipitate, or fibrinogen concentrate. Br J Anaesth. 2014;113:585–595.
Collins P, Abdul-Kadir R, Thachil J; Subcommittees on Women' s Health Issues in Thrombosis and Haemostasis and on Disseminated Intravascular Coagulation. Management of coagulopathy associated with postpartum hemorrhage: guidance from the SSC of the ISTH. J Thromb Haemost. 2016;14:205–10.
Collins PW, Cannings-John R, Bruynseels D, Mallaiah S, Dick J, Elton C et al. Viscoelastometric-guided early fibrinogen concentrate replacement during postpartum haemorrhage: OBS2, a double-blind randomized controlled trial. Br J Anaesth. 2017;119:411–21.
Cortet M, Deneux-Tharaux C, Dupont C, Colin C, Rudigoz RC, Bouvier-Colle MH, et al. Association between fibrinogen level and severity of postpartum haemorrhage: secondary analysis of a prospective trial. Br J of Anaesth. 2012;108:984–9.
Costa-Filho R, Hochleitner G, Wendt M, Teruya A, Spahn DR. Over 50 years of fibrinogen concentrate. Clin Appl Thromb Hemost. 2016;22:109–14.
Curry N, Foley C, Wong H, Mora A, Curnow E, Zarankaite A et al. Early fibrinogen concentrate therapy for major haemorrhage in trauma (E-FIT 1): results from a UK multi-centre, randomised, double blind, placebo-controlled pilot trial. Crit Care. 2018;22:164.
De Lorenzo C, Calatzis A, Welsch U, Heindl B. Fibrinogen concentrate reverses dilutional coagulopathy induced in vitro by saline but not by hydroxyethyl starch 6%. Anesth Analg. 2006;102:1194–1200.
Di Nisio M, Baudo F, Cosmi B, D'Angelo A, De Gasperi A, Malato A et al. Diagnosis and treatment of disseminated intravascular coagulation: guidelines of the Italian Society for Haemostasis and Thrombosis (SISET). Thromb Res. 2012;129:e177–84.
Downey LA, Andrews J, Hedlin H, Kamra K, McKenzie ED, Hanley FL et al. Fibrinogen concentrate as an alternative to cryoprecipitate in a postcardiopulmonary transfusion algorithm in infants undergoing cardiac surgery. Anesth Analg. 2019;doi:10.1213/ANE.0000000000004384.
European Association for the Study of the Liver. EASL Clinical Practical Guidelines on the management of acute (fulminant) liver failure. J Hepatol. 2017;66:1047–1081.
Fassl J, Lurati Buse G, Filipovic M, Reuthebuch O, Hampl K, Seeberger MD et al. Perioperative administration of fibrinogen does not increase adverse cardiac and thromboembolic events after cardiac surgery. Br J Anaesth. 2015;114:225–34.
Fenger-Eriksen C, Jensen TM, Kristensen BS, Jensen KM, Tonnesen E, Ingerslev J et al. Fibrinogen substitution improves whole blood clot firmness after dilution with hydroxyethyl starch in bleeding patients undergoing radical cystectomy: a randomized, placebo-controlled clinical trial. J Thromb Haemost. 2009;7:795–802.
Fominskiy E, Nepomniashchikh VA, Lomivorotov VV, Monaco F, Vitiello C, Zangrillo A et al. Efficacy and Safety of Fibrinogen Concentrate in Surgical Patients: A Meta-Analysis of Randomized Controlled Trials. J Cardiothorac Vasc Anesth. 2016;30:1196–204.
Franchini M, Lippi G. Fibrinogen replacement therapy: a critical review of the literature. Blood Transfus. 2012;10:23–7.
Fries D, Krismer A, Klingler A, Streif W, Klima G, Wenzel V et al. Effect of fibrinogen on reversal of dilutional coagulopathy: a porcine model. Br J Anaesth. 2005;95:172–7.
Fries D, Innerhofer P, Reif C, Streif W, Klingler A, Schobersberger W et al. The effect of fibrinogen substitution on reversal of dilutional coagulopathy: an in vitro model. Anesth Analg. 2006;102:347–51.
Frith D, Goslings JC, Gaarder C, Maegele M, Cohen MJ, Allard S et al. Definition and drivers of acute traumatic coagulopathy: clinical and experimental investigations. J Thromb Haemost. 2010;8:1919–25.
Galas F, Almeida JP, Fukushima JT, Vincent JL, Osawa EA, Zeferino S et al. Hemostatic effects of fibrinogen concentrate compared with cryoprecipitate in children after surgery: a randomised pilot trial. J Thorac Cardiovasc Surg. 2014;148:1647– 55.
Gayat E, Resche-Rigon M, Morel O, Possignol M, Mantz J, Nicolas-Robin A et al. Predictive factors of advanced interventional procedures in a multicentre severe postpartum haemorrhage study. Intensive Care Med. 2011;37:1816–25.
Giordano P, Luciani M, Grassi M, De Leonardis F, Coletti V, Santoro N. Supplementation of fibrinogen concentrate in children with severe acquired hypofibrinogenaemia during chemotherapy for acute lymphoblastic leukaemia: our experience. Blood Transfus. 2014;12:s156–7.
Groeneveld DJ, Adelmeijer J, Hugenholtz GC, Ariëns RA, Porte RJ, Lisman T. Ex vivo addition of fibrinogen concentrate improves the fibrin network structure in plasma samples taken during liver transplantation. J Thromb Haemost. 2015;13:2192–201.
Guasch E, Gilsanz F. Treatment of postpartum hemorrhage with blood products in a tertiary hospital: outcomes and predictive factors associated with severe hemorrhage. Clin Appl Thromb Hemost. 2016;22:685–92.
Haas T, Fries D, Velik-Salchner C, Reif C, Klingler A, Innerhofer P. The in vitro effects of fibrinogen concentrate, factor XIII and fresh frozen plasma on impaired clot formation after 60% dilution. Anesth Analg. 2008;106:1360–5.
Haas T, Spielmann N, Restin T, Seifert B, Henze G, Obwegeser J et al. Higher fibrinogen concentrations for reduction of transfusion requirements during major paediatric surgery: A prospective randomised controlled trial. Br J Anaesth. 2015;115:234–43.
Huissoud C, Carrabin N, Benchaib M, Fontaine O, Levrat A, Massingnon D et al. Coagulation assessment by rotation thrombelastometry in normal pregnancy. Thromb Haemost. 2009;101:755–61.
Idris SF, Hadjinicolaou AV, Sweeney M, Winthrop C, Balendran G, Besser M. The efficacy and safety of cryoprecipitate in the treatment of acquired hypofibrinogenaemia. Br J Haematol. 2014;166:458–61.
Innerhofer P, Fries D, Mittermayr M, Innerhofer N, von Langen D, Hell T et al. Reversal of trauma-induced coagulopathy using first-line coagulation factor concentrates or fresh frozen plasma (RETIC): a single-centre, parallel-group, open-label, randomised trial. Lancet Haematol. 2017;4:e258–71.
Itagaki Y, Hayakawa M, Maekawa K, Saito T, Kodate A, Honma Y et al. Early administration of fibrinogen concentrate is associated with improved survival among severe trauma patients: a single-centre propensity score-matched analysis. World J Emerg Surg. 2020;15:7. doi: 10.1186/s13017-020-0291-9.
Kaspereit F, Doerr B, Dickneite G. The effect of fibrinogen concentrate administration on coagulation abnormalities in a rat sepsis model. Blood Coagul Fibrinolysis. 2004;15:39–43.
Karkouti K, Callum J, Crowther MA, McCluskey SA, Pendergrast J, Tait G et al. The relationship between fibrinogen levels after cardiopulmonary bypass and large volume red cell transfusion in cardiac surgery: an observational study. Anesth Analg. 2013;117:14–22.
Karkouti K, Callum J, Rao V, Heddle N, Farkouh ME, Crowther MA, Scales DC. Protocol for a phase III, non-inferiority, randomised comparison of a new fibrinogen concentrate versus cryoprecipitate for treating acquired hypofibrinogenaemia in bleeding cardiac surgical patients: the FIBRES trial. BMJ Open. 2018:e020741. doi: 10.1136/bmjopen-2017-020741.
Karlsson M, Ternström L, Hyllner M, Baghaei F, Flinck A, Skrtic S et al. Prophylactic fibrinogen infusion reduces bleeding after coronary artery bypass surgery. A prospective randomised pilot study. Thromb Haemost. 2009;102:137–44.
Kelley W, Guzman N. Fresh Frozen Plasma (FFP). StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513347/ (accessed April 2019).
Kelly M, Waldhauser H, Vilchez T, Dlotko E, Hutcheson R. PSU8: Comparing costs of using fibrinogen concentrate versus fresh frozen plasma in the treatment of bleeding during cardiac surgery in the Netherlands. Value Heal. 2019;22:S893.
Kikuchi M, Itakura A, Miki A, Nishibayashi M, Ikebushi K, Ishihara O. Fibrinogen concentrate substitution therapy for obstetric hemorrhage complicated by coagulopathy. J Obstet Gynaecol Res. 2013;39:770–6.
Kindo M, Hoang Minh T, Gerelli S, Perrier S, Meyer N, Schaeffer M et al. Plasma fibrinogen level on admission to the intensive care unit is a powerful predictor of postoperative bleeding after cardiac surgery with cardiopulmonary bypass. Thromb Res. 2014;134:360–368.
Kozek-Langenecker SA, Ahmed AB, Afshari A, Albaladejo P, Aldecoa C, Barauskas G et al. Management of severe perioperative bleeding: guidelines from the European Society of Anaesthesiology: first update 2016. Eur J Anaesthesiol. 2017;34:332–95.
Kreuz W, Meili E, Peter-Salonen K, Haertel S, Devay J, Krzensk U et al. Efficacy and tolerability of a pasteurised human fibrinogen concentrate in patients with congenital fibrinogen deficiency. Transfus Apher Sci. 2005;32:247–53.
Kwapisz MM, Kent B, DiQuinzio C, LeGare J, Garnett S, Swyer W et al. The prophylactic use of fibrinogen concentrate in high‐risk cardiac surgery. Acta Anaesthesiol Scand. 2020;aas.13540.
Lancé MD, Ninivaggi M, Schols SE, Feijge MA, Oehrl SK, Kuiper GJ et al. Perioperative dilutional coagulopathy treated with fresh frozen plasma and fibrinogen concentrate: a prospective randomized intervention trial. Vox Sang. 2012;103:25–34.
Levi M, Toh CH, Thachil J, Watson HG. Guidelines for the diagnosis and management of disseminated intravascular coagulation. Br J Haematol. 2009;145:24–33.
Li J, Gong J, Zhu F, Moodie J, Newitt A, Uruthiramoorthy L et al. Fibrinogen concentrate in cardiovascular surgery: A meta-analysis of randomized controlled trials. Anesthesia & Analgesia. 2018;127:612–21.
Lunde J, Stensballe J, Wikkelsø A, Johansen M, Afshari A. Fibrinogen concentrate for bleeding--a systematic review. Acta Anaesthesiol Scand. 2014;58:1061–74.
Makino S, Takeda S, Kobayashi T, Murakami M, Kubo T, Hata T et al. National survey of fibrinogen concentrate usage for post-partum hemorrhage in Japan: Investigated by the Perinatology Committee, Japan Society of Obstetrics and Gynecology. J Obstet Gynaecol Res. 2015;41:1155–60.
Mallaiah S, Barclay P, Harrod I, Chevannes C, Bhalla A. Introduction of an algorithm for ROTEM-guided fibrinogen concentrate administration in major obstetric haemorrhage. Anaesthesia. 2015;70:166–175.
Manco-Johnson MJ, Dimichele D, Castaman G, Fremann S, Knaub S, Kalina U et al. Pharmacokinetics and safety of fibrinogen concentrate. J Thromb Haemost. 2009;7:2064–9.
Mathijssen IM. Guideline for care of patients with the diagnoses of craniosynostosis: working group on craniosynostosis. J Craniofac Surg. 2015;26:1735–807.
Matsunaga S, Takai Y, Nakamura E, Era S, Ono Y, Yamamoto K et al. The clinical efficacy of fibrinogen concentrate in massive obstetric haemorrhage with hypofibrinogenaemia. Sci Rep. 2017;7:46749. doi: 10.1038/srep46749.
McDonnell NJ. How to replace fibrinogen in postpartum haemorrhage situations? (Hint: Don't use FFP!). International Journal of Obstetric Anesthesia. 2018;33:4–7.
Mengoli C, Franchini M, Marano G, Pupella S, Vaglio S, Marietta M et al. The use of fibrinogen concentrate for the management of trauma-related bleeding: a systematic review and meta-analysis. Blood Transfus. 2017;15:318–24.
Mittermayr M, Streif W, Haas T, Fries D, Velik-Salchner C, Klingler A et al. Hemostatic changes after crystalloid or colloid fluid administration during major orthopedic surgery: the role of fibrinogen administration. Anesth Analg. 2007;105:905–17.
Najafi A, Shariat Moharari R, Orandi AA, Etezadi F, Sanatkar M, Khajavi MR. Prophylactic administration of fibrinogen concentrate in perioperative period of total hip arthroplasty: a randomized clinical trial study. Acta Med Iran. 2014;52:804–10.
Nascimento B, Goodnough LT, Levy JH. Cryoprecipitate therapy. Br J Anaesth. 2014;113:922–34.
Nascimento B, Callum J, Tien H, Peng H, Rizoli S, Karanicolas P et al. Fibrinogen in the initial resuscitation of severe trauma (FiiRST): a randomized feasibility trial. Br J Anaesth. 2016;117:775–782.
NICE guideline NG24. Transfusion: blood transfusion. Methods, evidence and recommendations. November 2015. Available at: https://www.ncbi.nlm.nih.gov/books/NBK327570/pdf/Bookshelf_NBK327570.pdf
Nienaber U, Innerhofer P, Westermann I, Schöchl H, Attal R, Breitkopf R et al. The impact of fresh frozen plasma vs coagulation factor concentrates on morbidity and mortality in trauma-associated haemorrhage and massive transfusion. Injury. 2011;42:697–701.
Noval-Padillo JA, León-Justel A, Mellado-Miras P, Porras-Lopez F, Villegas-Duque D, Gomez-Bravo MA et al. Introduction of fibrinogen in the treatment of hemostatic disorders during orthotopic liver transplantation: implications in the use of allogenic blood. Transplant Proc. 2010;42:2973–74.
Okerberg CK, Williams LA 3rd, Kilgore ML, Kim CH, Marques MB, Schwartz J et al. Cryoprecipitate AHF vs. fibrinogen concentrates for fibrinogen replacement in acquired bleeding patients - an economic evaluation. Vox Sang. 2016;111:292–8.
Peyvandi F. Results of an international, multicentre pharmacokinetic trial in congenital fibrinogen deficiency. Thromb Res. 2009;124:S9–11.
Poujade O, Zappa M, Letendre I, Ceccaldi PF, Vilgrain V, Luton D. Predictive factors for failure of pelvic arterial embolization for postpartum hemorrhage. Int J Gynaecol Obstet. 2012;117:119–23.
Rahe-Meyer N, Pichlmaier M, Haverich A, Solomon C, Winterhalter M, Piepenbrock S et al. Bleeding management with fibrinogen concentrate targeting a high-normal plasma fibrinogen level: a pilot study. Br J Anaesth. 2009;102:785–792.
Rahe-Meyer N, Hanke A, Schmidt DS, Hagl C, Pichlmaier M. Fibrinogen concentrate reduces postoperative bleeding when used as first-line hemostatic therapy during major aortic replacement surgery: results from a randomized, placebo-controlled trial. J Thorac Cardiovasc Surg. 2013a;145:S178–S185.
Rahe-Meyer N, Solomon C, Hanke A, Schmidt DS, Knoerzer D, Hochleitner G et al. Effects of fibrinogen concentrate as first-line therapy during major aortic replacement surgery: a randomized, placebo-controlled trial. Anesthesiology. 2013b;118:40–50.
Rahe-Meyer N, Levy JH, Mazer CD, Schramko A, Klein AA, Brat R et al. Randomized evaluation of fibrinogen vs placebo in complex cardiovascular surgery (REPLACE): a double-blind phase III study of haemostatic therapy. Br J Anaesth. 2016;117:41–51.
Rahe-Meyer N, Levy JH, Mazer CD, Schramko A, Klein AA, Brat R et al. Randomized evaluation of fibrinogen versus placebo in complex cardiovascular surgery: post hoc analysis and interpretation of phase III results. Interact Cardiovasc Thorac Surg. 2019;28:566–74.
Ranucci M, Baryshnikova E, Soro G, Ballotta A, De Benedetti D, Conti D. Multiple electrode whole-blood aggregometry and bleeding in cardiac surgery patients receiving thienopyridines. Ann Thorac Surg. 2011;91:123–129.
Ranucci M, Baryshnikova E, Crapelli GB, Rahe-Meyer N, Menicanti L, Frigiola A et al. Randomized, double-blinded, placebo-controlled trial of fibrinogen concentrate supplementation after complex cardiac surgery. J Am Heart Assoc. 2015;4:e002066.
Ranucci M, Pistuddi V, Baryshnikova E, Colella D, Bianchi P. Fibrinogen levels after cardiac surgical procedures: Association with postoperative bleeding, trigger values, and target values. Ann Thorac Surg. 2016;102:78–85.
Rourke C, Curry N, Khan S, Taylor R, Raza I, Davenport R et al. Fibrinogen levels during trauma hemorrhage, response to replacement therapy, and association with patient outcomes. J Thromb Haemost. 2012;10:1342–51.
Roy A, Sargent N, Rangarajan S, Alves S, Bell J, Stanford S et al. Fibrinogen concentrate vs cryoprecipitate in pseudomyxoma peritonei surgery: interim results from a prospective, randomized, controlled phase 2 study. Blood. 2018;132:2549.
Roy A, Stanford S, Nunn S, Alves S, Sargant N, Rangarajan S et al. Efficacy of fibrinogen concentrate in major abdominal surgery – A prospective, randomized, controlled study in cytoreductive surgery for pseudomyxoma peritonei. J Thromb Haemost. 2020;18:352–363.
Sabate A, Gutierrez R, Beltran J, Mellado P, Blasi A, Acosta F et al. Impact of preemptive fibrinogen concentrate on transfusion requirements in liver transplantation: A multicenter, randomized, double-blind, placebo-controlled trial. Am J Transplant. 2016;16:2421–9.
Sahin AS, Ozkan S. Treatment of obstetric hemorrhage with fibrinogen concentrate. Med Sci Monit. 2019;25:1814–21.
Schlimp CJ, Ponschab M, Voelckel W, Treichl B, Maegele M, Schöchl H. Fibrinogen levels in trauma patients during the first seven days after fibrinogen concentrate therapy: a retrospective study. Scand J Trauma Resusc Emerg Med. 2016;24:29.
Schöchl H, Nienaber U, Maegele M, Hochleitner G, Primavesi F, Steitz B et al. Transfusion in trauma: thromboelastometry-guided coagulation factor concentrate-based therapy versus standard fresh frozen plasma-based therapy. Crit Care. 2011;15:R83.
Seto S, Itakura A, Okagaki R, Suzuki M, Ishihara O. An algorithm for the management of coagulopathy from postpartum hemorrhage, using fibrinogen concentrate as first-line therapy. Int J Obstet Anaesth. 2017;32:11–16.
Simon L, Santi TM, Sacquin P, Hamza J. Pre-anaesthetic assessment of coagulation abnormalities in obstetric patients: usefulness, timing and clinical implications. Br J Anaesth. 1997;78:678–83.
Soleimani M, Masoumi N, Nooraei N, Lashay A, Safarinejad MR. The effect of fibrinogen concentrate on perioperative bleeding in transurethral resection of the prostate: a double-blind placebo-controlled and randomized study. J Thromb Haemost. 2017;15:255–62.
Solomon C, Pichlmaier U, Schoechl H, Hagl C, Raymondos K, Scheinichen D et al. Recovery of fibrinogen after administration of fibrinogen concentrate to patients with severe bleeding after cardiopulmonary bypass surgery. Br J Anaesth. 2010;104:555–62.
Solomon C, Cadamuro J, Ziegler B, Schöchl H, Varvenne M, Sørensen B et al. A comparison of fibrinogen measurement methods with fibrin clot elasticity assessed by thromboelastometry, before and after administration of fibrinogen concentrate in cardiac surgery patients. Transfusions. 2011;51:1695–796.
Solomon C, Schöchl H, Hanke A, Calatzis A, Hagl C, Tanaka K et al. Haemostatic therapy in coronary artery bypass graft patients with decreased platelet function: comparison of fibrinogen concentrate with allogeneic blood products. Scand J Clin Lab Invest. 2012;72:121–8.Sørensen B, Bevan D. A critical evaluation of cryoprecipitate for replacement of fibrinogen. British journal of haematology. 2010;149:834–43.
Spahn DR, Bouillon B, Cerny V, Duranteau J, Filipescu D, Hunt BJ et al. The European guideline on management of major bleeding and coagulopathy following trauma: fifth edition. Crit Care. 2019;23:98.
Stanworth SJ, Tinmouth AT. Plasma transfusion and use of albumin. In: Simon TL, editor. Rossi's Principles of Transfusion Medicine. 4th ed. Oxford, West Sussex, New Jersey: Blackwell Publishing; 2009:287–97.
Thachil J, Falanga A, Levi M, Liebman H, Di Nisio M, Scientific and Standardization Committee of the International Society on Thrombosis and Hemostasis. Management of cancer-associated disseminated intravascular coagulation: guidance from the SSC of the ISTH. J Thromb Haemost. 2015;13:671–5.
Theusinger OM, Baulig W, Seifert B, Emmert MY, Spahn DR, Asmis LM. Relative concentrations of haemostatic factors and cytokines in solvent/detergent‐treated and fresh‐frozen plasma. Br J Anaesth. 2011;106:505–11.
Tirotta CF, Lagueruela RG, Salyakina D, Wang W, Taylor T, Ojito J et al. Interval changes in ROTEM values during cardiopulmonary bypass in pediatric cardiac surgery patients. J Cardiothorac Surg. 2019;14.doi:10.1186/s13019-019-0949-0.
Wada H, Thachil J, Di Nisio M, Mathew P, Kurosawa S, Gando S et al. Guidance for diagnosis and treatment of DIC from harmonization of the recommendations from three guidelines. J Thromb Haemost. 2013;11:doi: 10.1111/jth.12155.
Wafaisade A, Lefering R, Maegele M, Brockamp T, Mutschler M, Lendemans S et al. Administration of fibrinogen concentrate in exsanguinating trauma patients is associated with improved survival at 6 hours but not at discharge. J Trauma Acute Care Surg. 2013;74:387–3.
Waldén K, Jeppsson A, Nasic S, Karlsson M. Fibrinogen concentrate to cardiac surgery patients with ongoing bleeding does not increase the risk of thromboembolic complications or death. Thromb Haemost. 2020;doi:10.1055/s-0039-3402759.
Wikkelsø AJ, Edwards HM, Afshari A, Stensballe J, Langhoff-Ross J, Albrechtsen C et al. Pre-emptive treatment with fibrinogen concentrate for postpartum haemorrhage: randomized controlled trial. Br J Anaesth. 2015;114:623–33.
Wong H, Curry N. Do we need cryoprecipitate in the era of fibrinogen concentrate and other specific factor replacement options? VOXS. 2018;13:23–28.
World Health Organization. Available at: http://whqlibdoc.who.int/publications/2010/9789241599375_eng.pdf (accessed 13 August 2019)
Yamamoto K, Usui A, Takamatsu J. Fibrinogen concentrate administration attributes to significant reductions of blood loss and transfusion requirements in thoracic aneurysm repair. Journal of Cardiothoracic Surgery. 2014;9:90.
Zadeh FJ, Janatmakan F, Soltanzadeh M, Zamankhani M. Investigating the effect of fibrinogen injection on bleeding in coronary artery bypass surgery: A clinical trial. Anesthesiol Pain Med. 2019;9:doi:10.5812/aapm.92165.
of interest
are looking at
saved
next event
Developed by EPG Health for Medthority in collaboration with CSL Behring, with content provided by CSL Behring.
Not intended for Healthcare Professionals outside Europe.