Fibrinogen Deficiency
Persistent fibrinogen deficiency can lead to bleeding complications and an increased risk in mortality. Early identification and treatment of fibrinogen deficiency could therefore save lives.
Correct perioperative management of fibrinogen loss experienced during heart surgery and postpartum haemorrhage is key to treatment success.
Fibrinogen role in haemostasis
Fibrinogen (Factor I) has many functions in haemostasis, a process that stops bleeding from damaged tissue. Such functions include coagulation (the creation of blood clots), fibrinolysis (the breaking down of blood clots), wound healing, inflammation and cell–cell interactions.
Role for fibrinogen in coagulation
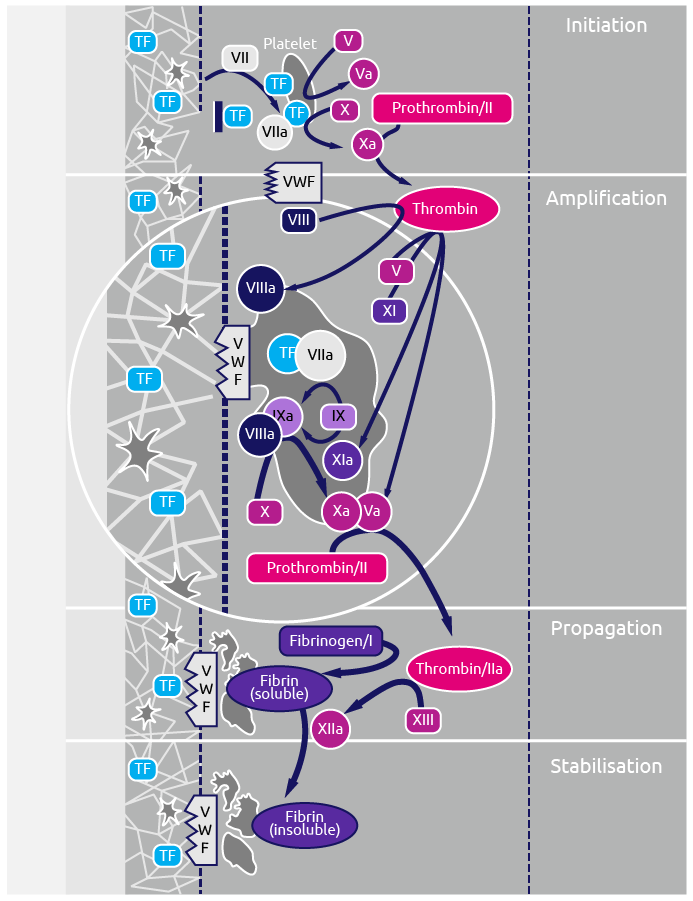
Coagulation, or blood clotting, is a highly regulated process that can be separated into four distinct stages (Monroe and Hoffman et al., 2007) (Figure 1):
- initiation; the sensing of damaged tissue
- amplification; signalling to factors required for clot formation
- propagation; initiating clot formation
- stabilisation; completing clot formation
Figure 1. The key stages of the coagulation cascade (Adapted from Monroe and Hoffman et al., 2007). TF, tissue factor; vWF, von Willebrand factor.
Fibrinogen plays an important structural role in both the propagation and stabilisation stages of coagulation, allowing for both clot formation and stability. An important feature of the coagulation process is its ability to trigger a fast response when tissue is damaged. As a result, fibrinogen is produced in an inactive state (like many other coagulation factors) by the liver and is secreted into blood to a normal variable range described as 2–4.5 g/L (Collins et al., 2014) or 1.5–3.5 g/L (Tennent et al., 2007; Neerman-Arbez & Casini, 2018).
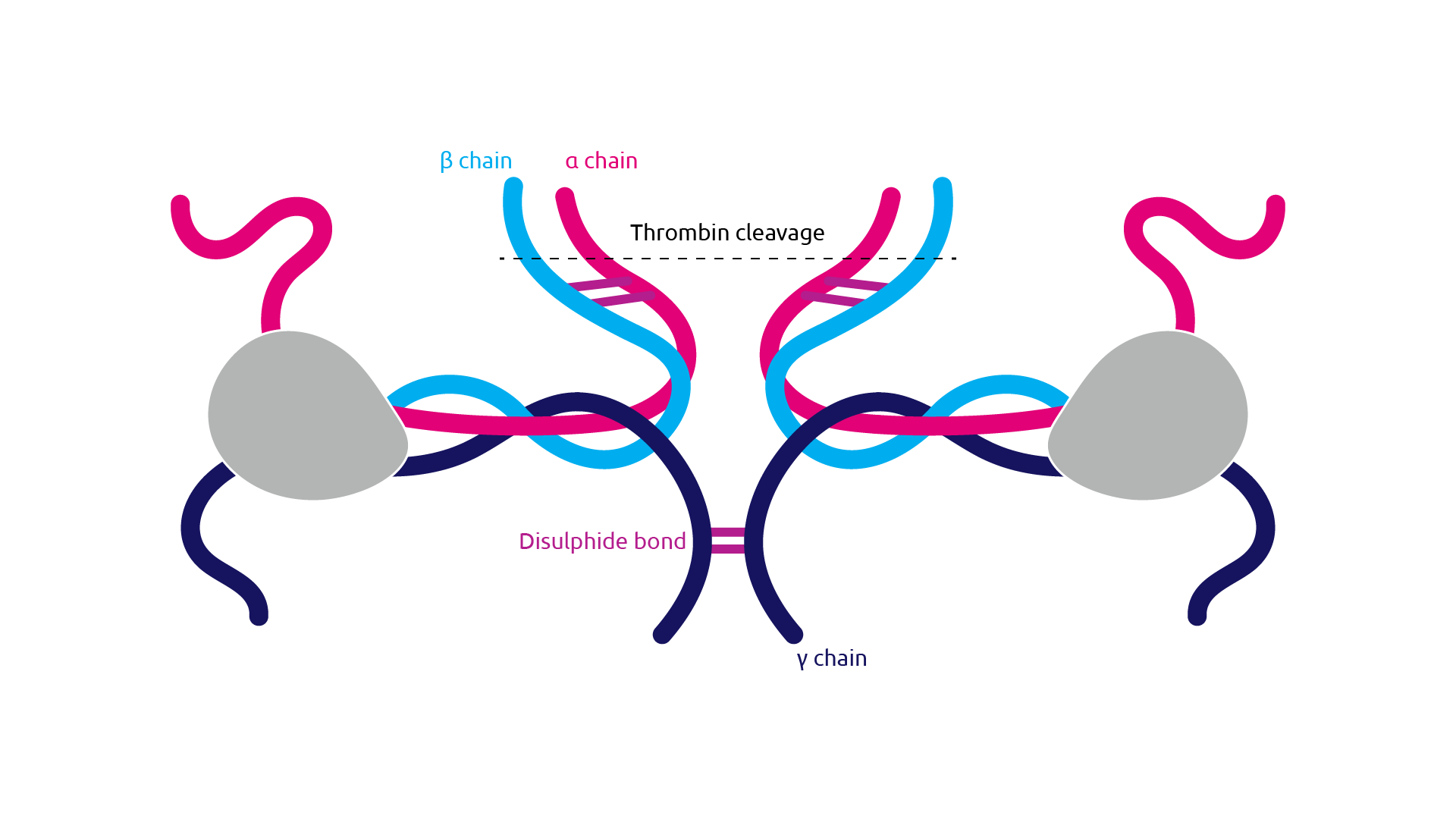
In its inactive state, fibrinogen is a large, soluble, hexameric glycoprotein that is made up of two copies each of three peptides Aα, Bβ and γ that are held together by disulphide bonds (Figure 2).
Figure 2. Fibrinogen structure; Purple = alpha chain, green = beta chain, red = gamma chain. (Adapted from Nacca et al., 2016).
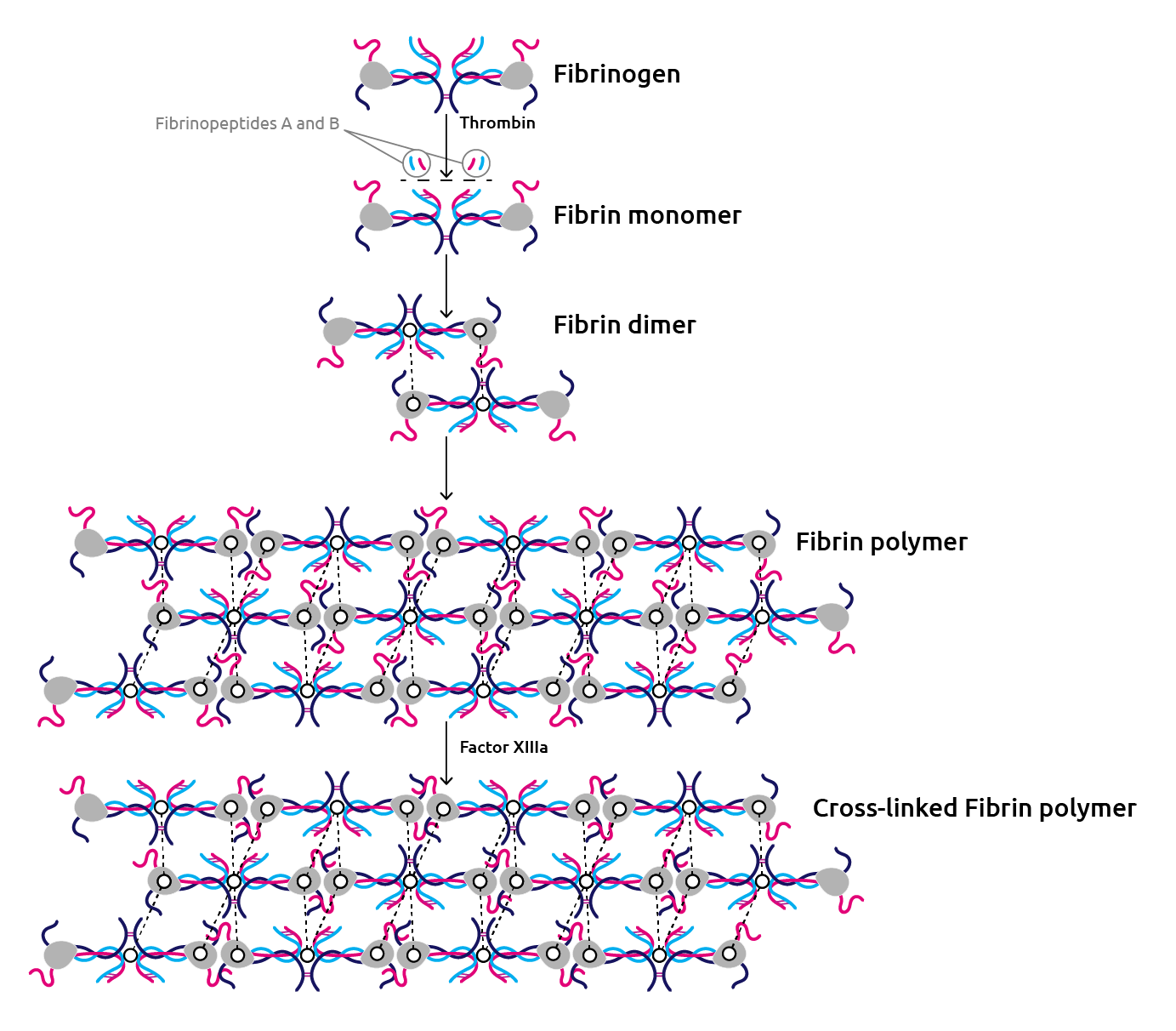
Upon initiation and amplification of the coagulation cascade (Figure 1), thrombin interacts with fibrinogen and cleaves the end-terminal regions of the Aα and Bβ peptides to produce fibrinopeptides A and B, respectively. This converts fibrinogen into soluble fibrin monomers that rapidly aggregate to form an insoluble fibrin polymer mesh. At the same time, thrombin converts another procoagulant, Factor XIII, into its active form, Factor XIIIa, a transglutaminase that cross-links glutamine residues from one fibrin monomer to lysine residues of another fibrin monomer, allowing for structural stability (Monroe and Hoffman et al., 2007) (Figure 3).
Figure 3. Fibrin polymers are stabilised by inter-monomer crosslinks catalysed by activated Factor XIII (Factor XIIIa) (Adapted from Adam et al., 2009).
Activated platelets aggregate on to the fibrin meshes through their surface glycoproteins IIb/IIIa and separately, bind to blood vessel walls to create a haemostatic plug (blood clot). Fibrin meshes also serve as a binding scaffold for other proteins involved in blood clotting (Laurens et al., 2006). This includes fibronectin (involved in cell adhesion), albumin (regulates fibrin ‘thickness’), thrombospondin (regulates the aggregation and binding of platelets to fibrin), Von Willebrand factor (involved in platelet adhesion) and fibrulin (promotes platelet adhesion to the extracellular matrix). Growth factors such as fibroblast growth factor-2 and vascular endothelial growth factor that encourage blood vessel repair also interact with fibrin meshes (Brown et al., 1996; Sahni et al., 1998). Finally, fibrin is involved in activating the immune response, the first line of defence upon tissue damage. This is achieved through fibrin binding and accumulating cells required for both an inflammatory response and for tissue repair (Esmon et al., 2012). Altogether, activated fibrinogen creates an adhesive and stable structure for building blood clots and promoting blood vessel repair.
Role for fibrinogen in clot degradation
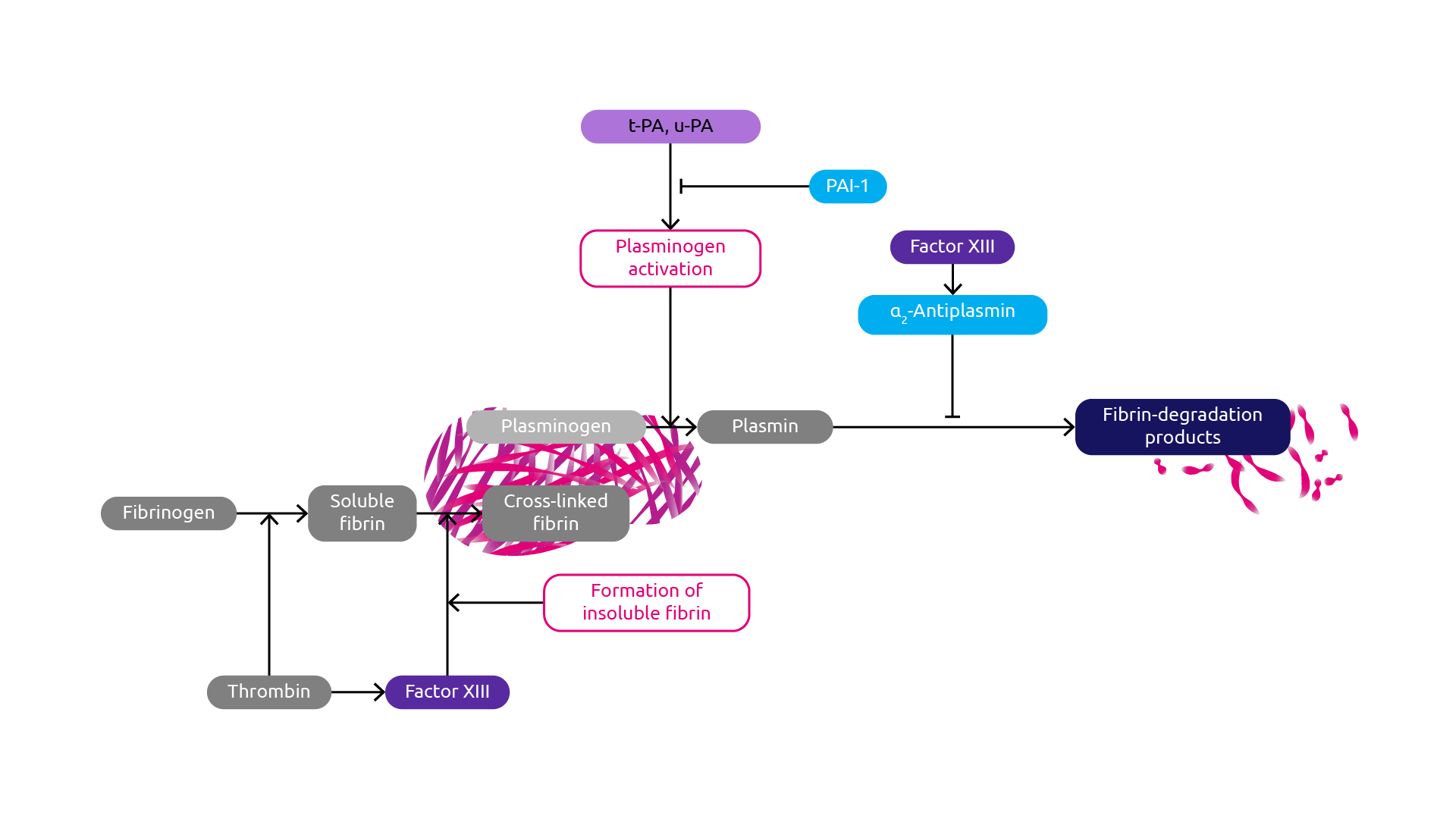
Fibrin is involved in regulating fibrinolysis, a process whereby blood clots that are no longer needed are broken down. This action reduces circulating blood clots that have the potential to block vessels and cause further damage (Chapin & Hajjar, 2015).
Plasmin is a serine protease enzyme that inactivates various blood plasma proteins through enzymatic cleavage (proteolysis), including fibrin clots. Plasmin is produced and released from the liver as a zymogen called plasminogen and two circulating types exist based on the number of glycosylation moieties they possess (one versus two). Type I plasminogen, which contains two glycosylation moieties, is preferentially recruited to blood clots. Freely circulating plasminogen is normally inactive until it binds to an activator such as a blood clot. This allows for a conformational change that promotes plasminogen conversion to plasmin through enzymes such as tissue plasminogen activator (tPA) (Chapin & Hajjar, 2015) (Figure 4). Importantly, fibrin is a cofactor that allows tPA activity and therefore is involved in promoting plasmin formation (Hoylaerts et al., 1982; Horrevoets et al., 1997). If plasmin levels are too low, as occurs in the absence of fibrin, then blood clots may persist as they are not being efficiently broken down, thus increasing the risk of thrombosis. Indeed, roles for fibrinogen in both clot formation and breakdown is ‘paradoxical’ in nature, whereby a deficiency can lead to reduced blood clotting and excessive bleeding, but at the same time, increases the risk of thrombotic complications.
Figure 4. Activated fibrinogen (fibrin) plays an important role in plasmin-dependent clot degradation (Adapted from Kohler & Grant, 2000). t-PA, tissue plasminogen activator; u-PA, urokinase plasminogen activator; PAI-1, plasminogen activator inhibitor-1.
Fibrinogen dysregulation
The structural role for fibrinogen in clot formation and the regulatory role of fibrin in fibrinolysis impacts greatly on haemostasis, particularly during trauma involving bleeding. This is highlighted in patients that have congenital deficiencies of fibrinogen who are prone to excessive bleeding. Furthermore, fibrinogen is the first coagulant to dramatically decrease as a result of fibrinogen consumption, dilution or bleeding during trauma, cardiac surgery and postpartum haemorrhage.
of interest
are looking at
saved
next event
Congenital fibrinogen deficiency
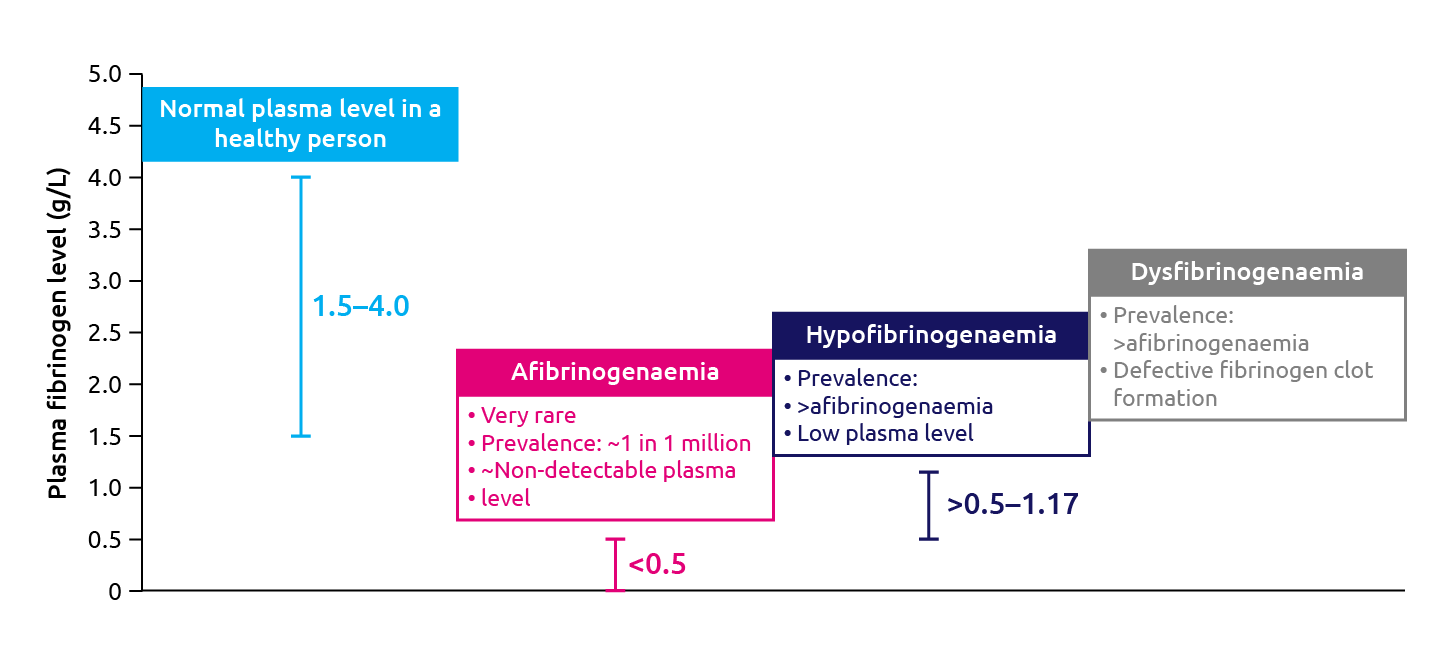
Low fibrinogen levels and quality impacts negatively on blood clot formation and can lead to excessive bleeding and death if left untreated. Fibrinogen deficiencies can either be inherited (congenital fibrinogen deficiency) or acquired (acquired fibrinogen deficiency). Congenital fibrinogen deficiency manifests from no detectable fibrinogen (such as for congenital afibrinogenaemia) to some detectable fibrinogen (such as for congenital hypofibrinogenaemia) (Figure 5). It is usually the result of a heterozygous or homozygous mutation in one of the three fibrinogen genes (FGA, FGB and FGG) that are located on the long arm of chromosome 4 (4q31.3). Key symptoms include uncontrollable bleeding and thrombolytic complications. The onset of bleeding can be spontaneous, or as a result of both minor and major traumas. Bleeding phenotypes can range from being asymptomatic to severe and is partially dependent on both the level and quality of fibrinogen available (Peyvandi et al., 2012). There are two types of congenital deficiency:
- Type I: quantitative defect
- Type 2: qualitative defect
Figure 5. Fibrinogen levels in congenital fibrinogen deficiencies (Reviewed in Peyvandi et al., 2012).
Type I: Congenital Afibrinogenaemia
Congenital afibrinogenaemia is a rare inherited bleeding disorder that affects approximately 1:1,000,000 people (Tziomalos et al., 2009). It is the result of one or more recessive mutations in any of the genes that encode the three peptides (Aα, Bβ and γ) that constitute the fibrinogen hexamer. Over 245 mutations have been identified to date, each affecting either the synthesis, stability or secretion of fibrinogen (Asselta et al., 2006). Consequently, people who suffer from congenital afibrinogenaemia have almost no detectable plasma fibrinogen (less than 0.1 g/L). They are therefore unable to form blood clots normally and are at greater risk of thrombolytic complications.
Afibrinogenaemia manifests clinically as minimal bleeding to serious haemorrhage and is usually diagnosed at birth, when bleeding from the umbilical cord is difficult to stop. Other symptoms throughout life include reduced wound healing, nose bleeds and bleeding of joints, mucous membranes, the central nervous system and the gastrointestinal tract. Additionally, women sufferers experience prolonged and heavy menstruation (menorrhagia) and can experience spontaneous abortions, usually at 5 to 8 weeks of gestation, as well as postpartum haemorrhage.
Despite having a lower capacity to produce blood clots, afibrinogenaemia patients can also experience spontaneous thrombosis in peripheral arteries and in cerebral and hepatic veins (Lak et al., 1999; Oruc et al., 2006; Fuchs et al., 2007). This could be due to reduced fibrinolysis of present clots or reduced thrombin sequestration by fibrinogen. For the latter, patients that lack fibrinogen tend to display increased thrombin levels, with a subsequent increase in prothrombin activation (de Bosch et al., 2002; Mosesson et al., 2003). Further, free thrombin may stimulate platelets to release several growth factors that impact on vascular smooth muscle proliferation and blood vessel thickness (Korte et al., 1994), altogether leading to increased thrombolytic complications.
Type I: Congenital Hypofibrinogenaemia
Hypofibrinogenaemia describes a partial fibrinogen deficiency with plasma levels below 1.5 g/L (de Moerloose et al., 2013; Neerman et al., 2018). The partial deficiency is mostly the result of a heterozygous mutation in the genes that encode the fibrinogen peptides. Patients of hypofibrinogenaemia tend to be asymptomatic and are usually diagnosed during some form of intervention, for instance through pre-operative testing or through major trauma when excessive bleeding is evident (Casini et al., 2016). Female sufferers are also more likely to suffer pregnancy loss and postpartum haemorrhage (Hill et al., 2006). In rare cases, liver disease may arise as a result of aggregate accumulation in the endoplasmic reticulum of hepatocytes (Brennan et al., 2000; Brennan et al., 2002; Dib et al., 2007; Brennan et al., 2010).
Type II: Dysfibrinogenaemia
Dysfibrinogenaemia is a rare (15 per 100,000) autosomal dominant disorder caused mostly by heterozygous missense mutations in any one of the three genes that encode the fibrinogen peptides. Whilst there are over 100 nucleotide variations, two key mutations account for 75% of dysfibrinogenaemia cases (FGA: Arg35 and FGG: Arg301) (GEHT database). These mutations impact on the quality of fibrinogen produced rather than the quantity. There are two known mechanisms of quality defects; either fibrinogen is unable to bind thrombin, or the resulting fibrin clot cannot undergo plasmin degradation (de Moerloose et al., 2013).
Like congenital hypofibrinogenaemia, dysfibrinogenaemia patients tend to be asymptomatic, however display bleeding and thrombolytic complications upon trauma. In the absence of genetic family history, diagnosis is therefore usually made accidentally because of unusual coagulation test results, characterised by a ratio of functional activity to antigen level of approximately 0.7 or less (Casini et al., 2015).
Type II: Hypodysfibrinogenaemia
Hypodysfibrinogenaemia is the rarest of the congenital fibrinogen disorders. Patients who suffer hypodysfibrinogenaemia present low-quality fibrinogen at reduced levels. As such, these patients share features with both hypofibrinogenaemia and dysfibrinogenaemia patients. There are at least 32 known contributor mutations in the three genes that encode the fibrinogen peptides (Casini et al., 2017). Symptoms range from being mostly asymptomatic to experiences of severe bleeds in cases of trauma, such as postpartum haemorrhage, and recurrent thrombosis.
Whilst there aren’t many reported cases of hypodysfibrinogenaemia, functional fibrinogen levels were found to be in the range of 0.1–1.1 g/L whereas antigenic fibrinogen levels were higher, at 0.31–1.8 g/L. The mean ratio of fibrinogen functional activity/antigenic levels was 0.46 (range of 0.07–1.25) (Casini et al., 2017). Indeed, hypodysfibrinogenaemia patients are often misdiagnosed as having hypofibrinogenaemia based on the fibrinogen level results.
Other forms of fibrinogen deficiency
Acquired fibrinogen deficiency is far more common in the general population and can arise as a result of consumptive and dilutional coagulopathy and due to blood loss experienced during major trauma, postpartum haemorrhage and cardiac surgery.
of interest
are looking at
saved
next event
Acquired fibrinogen deficiency
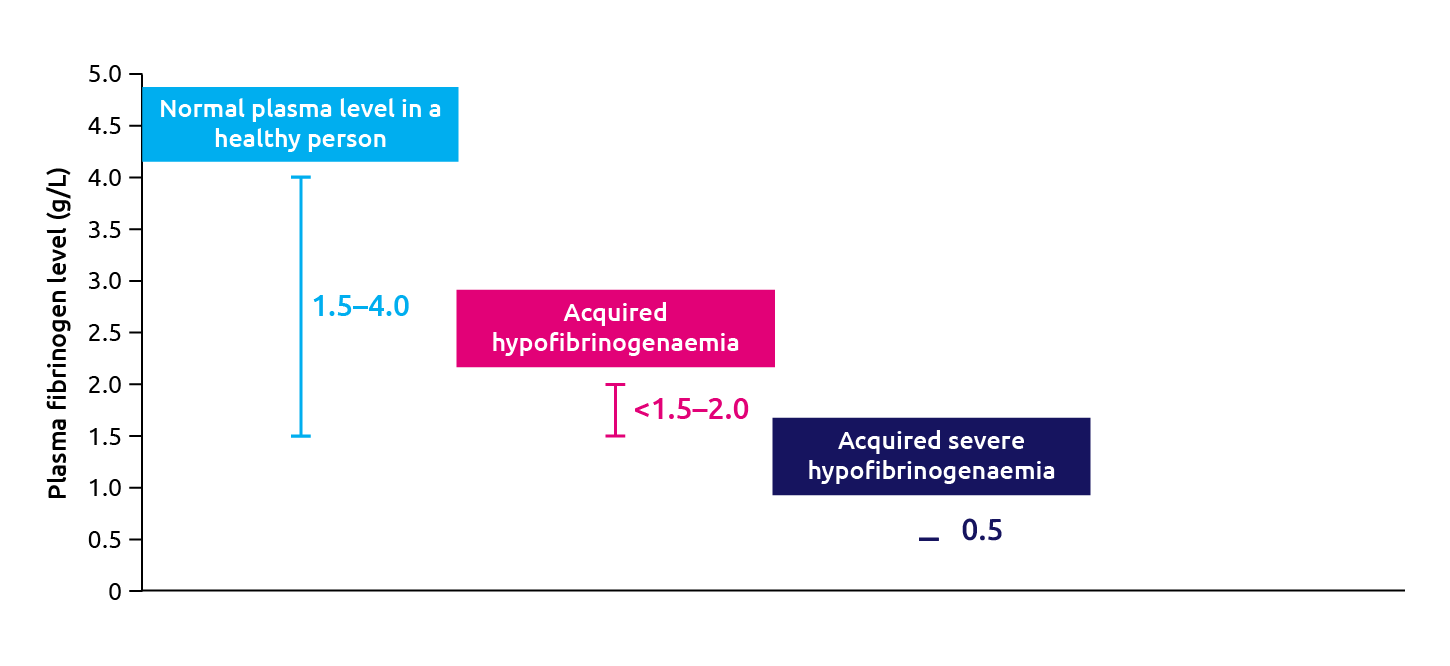
Acquired hypofibrinogenaemia is much more common than congenital fibrinogen deficiency and is defined by a fibrinogen level of less than 1.5 g/L (Figure 6).
Figure 6: Fibrinogen levels in acquired fibrinogen deficiencies (Reviewed in Besser et al., 2016; Kozek-Langemecker et al., 2017).
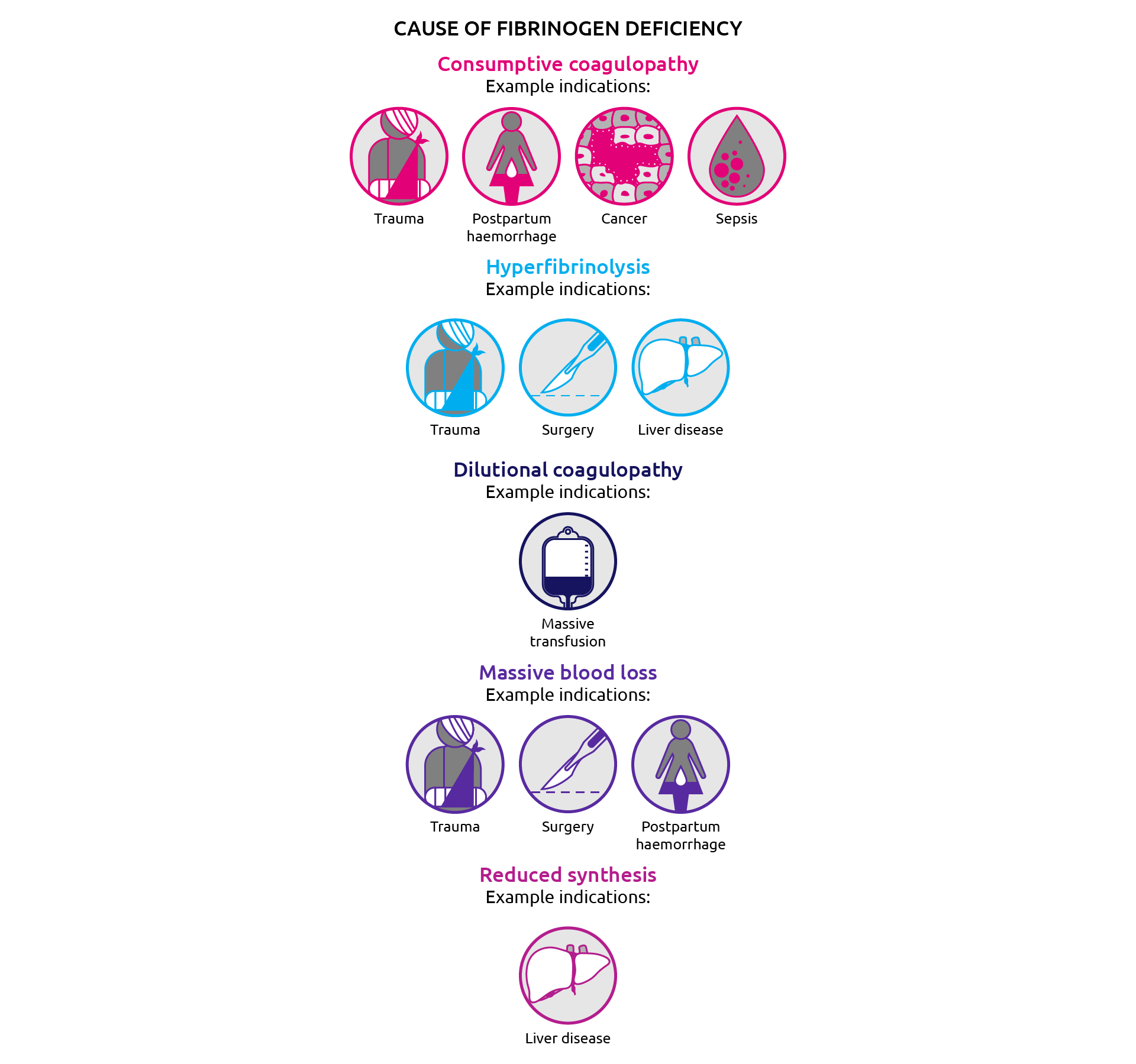
Common causes of acquired fibrinogen deficiency include excessive blood loss (for example during major trauma, surgery and postpartum haemorrhage), haemodilution (during massive transfusion), consumptive coagulopathies (disseminated intravascular coagulation and hyperfibrinolysis) and end stage liver disease (Fries & Martini, 2010; Besser & MacDonald, 2016) (Figure 7).
Figure 7. Acquired hypofibrinogenaemia (Reviewed in Franchini & Lippi, 2012; Besser et al., 2016).
Acquired fibrinogen deficiency is a lot more complex than congenital fibrinogen deficiency. Congenital fibrinogen deficiency is associated directly with defects in fibrinogen protein synthesis, secretion or quality whilst other coagulation factors function normally. In acquired fibrinogen deficiency, fibrinogen is often associated with multiple factor deficiencies as well as pathophysiologic conditions such as shock, fibrinolysis, hypothermia and acidosis (Maegele et al., 2017). Indeed, traumatic coagulopathy (TC) together with acidosis and hypothermia is often referred to as the ‘lethal triad’ of death which is further aggravated by further accumulation of coagulation factor deficits, termed trauma induced coagulopathy (TIC) (Maegele et al., 2017). Diagnosis and treatment of acquired fibrinogen deficiency is subsequently more complex and difficult.
Consumptive coagulopathy
Consumptive coagulopathy, sometimes referred to as disseminated intravascular coagulation (DIC), is usually associated with an unusually high level of activation in the coagulation system and involves dysregulation of both procoagulants and anticoagulants. The outcome is the depletion of factors required for coagulation completion, one of which is fibrinogen. The measurable outcome of DIC varies according to the trigger factor, which can include sepsis, cancer, trauma and hepatic or obstetric disorders. It is important to note that in some scenarios, fibrinogen levels may remain in the normal range; however, it is unable to play its role in coagulation as a result of other consumptive complications related to DIC (Venugopal, 2014). It is therefore important to measure not only fibrinogen levels, but also other markers of clot formation in the diagnosis of DIC.
Hyperfibrinolysis
Fibrinolysis is the normal process for clearing blood clots once they are no longer needed. In the case of hyperfibrinolysis, blood clots are cleared too efficiently, thus preventing adequate platelet aggregation and clot establishment. It is often caused by high plasmin levels, a key factor responsible for the degradation of fibrin. Hyperfibrinolysis leads to increased bleeding and has been observed in patients of liver disease, trauma and surgical procedures (Teufelsbauer et al., 1992; Palmer et al., 1995; Ferro et al., 2009; Theusinger et al., 2011).
Dilutional coagulopathy
Extensive fluid replacement with red blood cells and intravenous fluids that do not have platelets or coagulant factors can lead to the dilution of both pro- and anticoagulants. This leads to a loss of coagulation regulation and is frequently observed with massive transfusions. The outcome is the onset of thrombocytopenia (low blood platelet count) and fibrinogen levels below 1.5 g/L after a 1–1.5 blood volume replacement (JPAC Transfusion Handbook, 2014).
Blood loss
A major haemorrhage can be defined by speed of bleeding (150 mL/minute) and volume of blood lost; either one blood volume (approximately 70 mL/kg in an adult) within 24 hours or 50% total volume in less than 3 hours. It is characterised by a systolic blood pressure of less than 90 mm Hg or a heart rate of more than 110 beats/minute (JPAC Transfusion Handbook, 2014). Notably, the amount and cause of bleeding is influenced by various patient-specific factors and diagnosis must be interpreted in the context of the individual clinical scenario (JPAC Transfusion Handbook, 2014). The importance of fibrinogen levels in major blood loss has been extensively studied, particularly in cardiac surgery and postpartum haemorrhaging.
of interest
are looking at
saved
next event
Cardiac surgery
Major bleeding during cardiac surgery is an outcome that warrants the use of blood transfusions and reinterventions, both of which are risk factors for mortality (Moulton et al., 1996; Makar et al., 2010; Vivacqua et al., 2011). Acute coagulation defects are a common complication in cardiac surgery and can arise as a result of tissue injury and hypotension, with haemodilution, hyperfibrinolysis, and inflammatory responses all helping to sustain bleeding (Levy et al., 2005; Karkouti et al., 2010; Makar et al., 2010; Chee et al., 2016). An added complication is that this patient group are likely to have comorbidities and ongoing anticoagulant treatment for the prevention of heart disease, or anticoagulants administered during surgery to prevent clot formation (such as heparin) (Scrutinio & Giannuzzi, 2008).
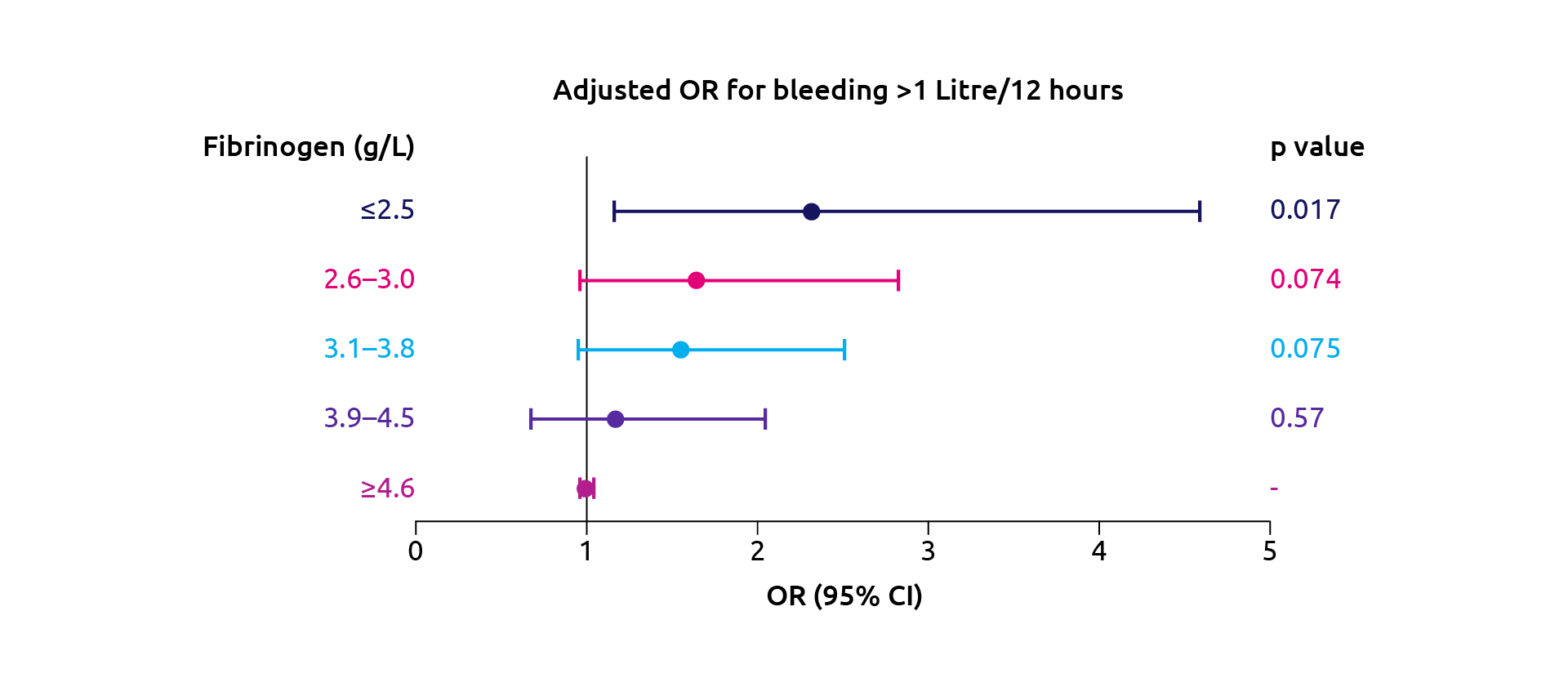
Many studies have found that both preoperative and postoperative fibrinogen levels are negatively correlated with blood loss during cardiac surgery (Kozek-Langenecker et al., 2017). In one study of 1,954 patients undergoing cardiac surgery, a preoperative plasma fibrinogen level of less than 2.5 g/L was independently associated with increased risk of postoperative bleeding when compared with a preoperative plasma fibrinogen level of 4.6 g/L or more (Walden et al., 2014) (Figure 8).
Figure 8. Association of preoperative plasma fibrinogen levels and risk of postoperative bleeding (adapted from Walden et al., 2014). OR, odds ratio; CI, confidence interval.
A systematic review and meta-analysis of 20 research articles covering a total of 5,972 cardiac surgery patients indicated that low pre- and postoperative fibrinogen levels are associated with greater blood loss in cardiac surgery (Gielen et al., 2014). The authors identified a significant negative pooled correlation between:
- postoperative blood loss and preoperative fibrinogen levels (r= -0.40; 95% confidence interval −0.58 to −0.18)
- postoperative blood loss and postoperative fibrinogen levels (r= -0.23; 95% confidence interval −0.29 to −0.16)
Some studies focus on distinct clinical settings. For instance, Liu et al. carried out a retrospective study of 125 patients with type A acute aortic dissection, a tear in the inner layer of the ascending aorta that redirects blood to create a false lumen (Liu et al., 2018). The findings indicated that a fibrinogen level of less than 2.17 g/L was independently associated with higher in-hospital mortality in these patients (adjusted odds ratio 5.527; 95% confidence interval 1.660–18.401; p=0.005).
Figure 9. Association of fibrinogen level and in-hospital mortality (adapted from Liu et al., 2018).
Low postoperative fibrinogen is also a risk factor for excessive bleeding following cardiopulmonary bypass (CPB). In a prospective, multi-centre observational cohort study of cardiac surgery patients undergoing CPB (n = 1,956), plasma fibrinogen levels measured at the time of ICU admission was correlated with excessive bleeding, defined as the 24–hour chest tube output exceeding the 90th percentile of the distribution (n = 189) (Kindo et al., 2014). Fibrinogen levels were 2.5 g/L for the control group compared to 2.1 g/L for the group that experienced excessive bleeding (p<0.0001). On admission to intensive care, there was a significant negative correlation between fibrinogen levels and excessive bleeding (r = -0.237; p<0.0001). In this study, the authors recommend a cut-off of 2.2 g/L fibrinogen level at the point of admission to intensive care for predicting postoperative bleeding (Kindo et al., 2014).
of interest
are looking at
saved
next event
Postpartum haemorrhage
Postpartum haemorrhage (PPH) is a bleeding complication that accounts for approximately one in four maternal deaths, making it the most common cause of maternal death worldwide (Kassebaum et al., 2014). The incidence rate in developed countries has increased in recent years, related primarily to an increase in the use of oxytocin for progressing labour (Belghiti et al., 2011). PPH is usually the result of uterine atony, but may be due to uterine rupture, abnormal placentation, placental abruption, genital tract trauma and coagulation defects (Abdul-Kadir et al., 2014). Comorbidities such as foetal death can also impact on PPH severity.
During pregnancy, several haemostatic changes induce a prothrombotic state, with an increase in fibrinogen and procoagulation factors, and a decrease in anticoagulants (O'Riordan & Higgins, 2003; Brenner, 2004). This implies an inherent importance for hypercoagulability in women giving birth, supported further by the fact that both primary and secondary coagulopathies are risk factors for PPH (Cortet et al., 2012).
Fibrinogen levels increase during pregnancy by about 2 g/L overall, to an average of 5 g/L at nine months (Simon et al., 1997; Huissoud et al., 2009). As with other traumas involving major blood loss, fibrinogen is one of the first coagulants to drastically decrease in PPH (Chauleur et al., 2008; de Lloyd et al., 2011). Indeed, this loss may be due to both dilutional and consumptive coagulopathies such as DIC and hyperfibrinolysis (Roberts et al., 2018).
In the last decade, studies have been documenting the correlation between fibrinogen levels and PPH severity and the role for fibrinogen as a diagnostic indicator. In an early study, a fibrinogen level of less than 2 g/L, measured at the point of uterotonic treatment, was found to be 100% predictive of severe PPH. The same study showed that each 1 g/L decrease in fibrinogen leads to an approximately 2.6-fold increase in risk for severe PPH (Charbit et al., 2007). This finding was reiterated in the 2012 Cortet study, in which fibrinogen levels were correlated to the severity of PPH in women who took part in the PITHAGORE6 trial (Deneux-Tharax et al., 2010; Cortet et al., 2012). Women with PPH who did not progress to severe PPH had a mean fibrinogen level of 4.2 g/L versus the 3.4 g/L measured in those who experienced severe PPH (p<0.001). Fibrinogen level was associated to PPH severity independently of other factors with an odds ratio of 1.9 for fibrinogen levels of 2–3 g/L, and 11.99 for fibrinogen <2 g/L.
In a more recent prospective study of 809 patients who underwent vaginal deliveries, the difference in predelivery fibrinogen levels for PPH and severe PPH was slightly less but still significant (4.67 g/L and 4.22 g/L, respectively; p=0.004; where incidence rates were 12% and 3.5%) (Niepraschk-von Dollen et al., 2016).
Other risk factors for the progression of PPH to severe PPH include increasing age and obstetric comorbidities such as foetal death, myomas and placenta previa. Only fibrinogen levels are modifiable in a clinical setting and therefore fibrinogen monitoring should be standardised for patients who present additional non–modifiable risk factors (Guasch & Gilsanz, 2016).
Fibrinogen levels are easily modifiable in practice. Fibrinogen monitoring should therefore be standard practice for patients that present comorbidities.
Whilst further investigation is required to understand fully whether low fibrinogen levels are causative in the onset of PPH, there is strong evidence that fibrinogen levels serve as an early diagnostic marker for PPH severity. Given that fibrinogen is critical for clot formation in coagulation, PPH patients are often treated with fibrinogen replacement therapies. This will be discussed more extensively in the treatment section.
Liver disease
Abnormal fibrinogen function is common in most patients of cirrhosis, chronic active liver disease and hepatic failure. End stage liver disease (ESLD) can negatively impact on fibrinogen synthesis and metabolism. ESLD patients tend to have normal to high levels of plasma fibrinogen; however, there are scenarios where ESLD patients present either high levels of fibrinogen degradation products indicating hyperfibrinolysis or acquired hypofibrinogenaemia with fibrinogen levels of less than 0.8–1.0 g/L (Saner & Kirchner, 2016).
of interest
are looking at
saved
next event
of interest
are looking at
saved
next event
Fibrinogen deficiency references
Abdul‐Kadir R, McLintock C, Ducloy A, El‐Refaey H, England A, Federici AB, et al. Evaluation and management of severe PPH. Transfusion. 2014;54:1756–68.
Adam SS, Key NS, Greenberg CS. D-dimer antigen: current concepts and future prospects. Blood. 2009;113;2878–87.
Asselta R, Duga S, Tenchini ML. The molecular basis of quantitative fibrinogen disorders. J Thromb Haemost. 2006;4(10):2115–29.
Belghiti J, Kayem G, Dupont C, Rudigoz R, Bouvier-Colle M, Deneux-Tharaux C et al. Oxytocin during labour and risk of severe postpartum haemorrhage: a population-based, cohort-nested case-control study. BMJ Open. 2011;1:e000514.
Besser MW, MacDonald SG. Acquired hypofibrinogenemia: current perspectives. J Blood Med. 2016;7:217–25.
Brennan SO, Wyatt J, Medicina D, Callea F, George PM. Fibrinogen brescia: hepatic endoplasmic reticulum storage and hypofibrinogenemia because of a gamma284 Gly—>Arg mutation. Am J Pathol. 2000;157:189–96.
Brennan SO, Maghzal G, Shneider BL, Gordon R, Magid MS, George PM. Novel fibrinogen gamma375 Arg—>Trp mutation (fibrinogen aguadilla) causes hepatic endoplasmic reticulum storage and hypofibrinogenemia. Hepatology. 2002;36:652–58.
Brennan SO, Davis RL, Conard K, Savo A, Furuya KN. Novel fibrinogen mutation γ314Thr→Pro (fibrinogen AI duPont) associated with hepatic fibrinogen storage disease and hypofibrinogenaemia. Liver Int. 2010;30:1541–47.
Brenner B. Haemostatic changes in pregnancy. Thrombosis Research. 2004;114:409–14.
Brown KJ, Maynes SF, Bezos A, Maguire DJ, Ford MD, Parish CR. A novel in vitro assay for human angiogenesis. Lab Invest. 1996;75:539–55.
Casini A, Neerman-Arbez M, Ariens RA, de Moerloose P. Dysfibrinogenemia: from molecular anomalies to clinical manifestations and management. J Thromb Haemost. 2015;13:909–19.
Casini A, de Moerloose P, Neerman-Arbez M. Clinical features and management of congenital fibrinogen deficiencies. Semin Thromb Hemost. 2016;42:366–74.
Casini A, Brungs T, Lavenu‐Bombled C, Vilar R, Neerman‐Arbez M, Moerloose P. Genetics, diagnosis and clinical features of congenital hypodysfibrinogenemia: a systematic literature review and report of a novel mutation. J Thromb Haemost. 2017;15:876–88.
Chapin JC, Hajjar KA. Fibrinolysis and the control of blood coagulation. Blood Rev. 2015;29:17–24.
Charbit B, Mandelbrot L, Samain E, Baron G, Haddaoui B, Keita H et al. The decrease of fibrinogen is an early predictor of the severity of postpartum haemorrhage. J Thromb Haemost. 2007;5:266–73.
Chauleur C, Cochery-Nouvellon E, Mercier E, Aya G, Fabbro-Peray P, Mismetti P et al. Some hemostasis variables at the end of the population distributions are risk factors for severe postpartum hemorrhages. J Thromb Haemost. 2008;6:2067–74.
Chee YE, Liu SE, Irwin MG. Management of bleeding in vascular surgery. British Journal of Anaesthesia. 2016;117:ii85–ii94.
Collins PW, Solomon C, Sutor K, et al. Theoretical modelling of fibrinogen supplementation with therapeutic plasma, cryoprecipitate, or fibrinogen concentrate. Br J Anaesth. 2014;113:585–95.
Cortet M, Deneux-Tharaux C, Dupont C, Colin C, Rudigoz RC, Bouvier-Colle MH, et al. Association between fibrinogen level and severity of postpartum haemorrhage: secondary analysis of a prospective trial. Br J of Anaesth. 2012;108:984–9.
de Bosch NB, Mosesson MW, Ruiz-Saez A, Echenagucia M, Rodriguez-Lemoin A. Inhibition of thrombin generation in plasma by fibrin formation (Antithrombin I). Thromb Haemost. 2002;88:253–58.
de Lloyd L, Bovington R, Kaye A, Collis RE, Rayment R, Sanders J et al. Standard haemostatic tests following major obstetric haemorrhage. International Journal of Obstetric Anesthesia. 2011;20:135–41.
de Moerloose P, Casini A, Neerman-Arbez M. Congenital fibrinogen disorders: an update. Semin Thromb Hemost. 2013;39:585–95.
Deneux-Tharaux C, Dupont C, Colin C, Rabilloud M, Touzet S, Lansac J et al. Multifaceted intervention to decrease the rate of severe postpartum haemorrhage: the PITHAGORE6 cluster-randomised controlled trial. BJOG. 2010;117:1278–87.
Dib N, Quelin F, Ternisien C, Hanss M, Michalak S, De Mazancourt P et al. Fibrinogen angers with a new deletion (gamma GVYYQ 346-350) causes hypofibrinogenemia with hepatic storage. J Thromb Haemost. 2007;5:1999–2005.
Esmon CT, Xu J, Lupu F. Innate immunity and coagulation. J Thromb Haemost. 2011;9:182–8.
Ferro D, Celestini A, Violi F. Hyperfibrinolysis in liver disease. Clin Liver Dis. 2009;13:21–31.
Franchini M, Lippi G. Fibrinogen replacement therapy: a critical review of literature. Blood Transfus. 2012;10:23–27.
Fries D, Martini WZ. Role of fibrinogen in trauma-induced coagulopathy. British Journal of Anaesthesia. 2010;105:116–21.
Fuchs RJ, Levin J, Tadel M, Merritt W. Perioperative coagulation management in a patient with afibrinogenemia undergoing liver transplantation. Liver Transpl. 2007;13:752–56.
Gielen C, Dekkers O, Stijnen T, Schoones J, Brand A, Klautz R et al. The effects of pre- and postoperative fibrinogen levels on blood loss after cardiac surgery: a systematic review and meta-analysis. Interact Cardiovasc Thorac Surg. 2014;18:292–8.
Guasch E, Gilsanz F. Treatment of postpartum haemorrhage with blood products in a tertiary hospital: outcomes and predictive factors associated with severe haemorrhage. Clin Appl Thromb Hemost. 2016;22:685–92.
Hill MB, Brennan SO, Dear A, Strong J, Nejim T, Dolan G. Fibrinogen Nottingham II: a novel Bbeta Arg264gly substitution causing hypofibrinogenaemia. Thromb Haemost. 2006;96:378–80.
Hoffman M, Monroe DM. Coagulation 2006: a modern view on hemostasis. Hematol Oncol Clin North Am. 2007;21:1–11.
Horrevoets AJ, Pannekoek H, Nesheim ME. A steady-state template model that describes the kinetics of fibrin-stimulated [Glu1]- and [Lys78] plasminogen activation by native tissue-type plasminogen activator and variants that lack either the finger or kringle-2 domain. J Biol Chem. 1997;272:2183–91.
Hoylaerts M, Rijken DC, Lijnen HR, Collen D. Kinetics of the activation of plasminogen by human tissue plasminogen activator. Role of fibrin. J Biol Chem. 1982;257:2912–19.
Huissoud C, Carrabin N, Benchaib M, Fontaine O, Levrat A, Massingnon D et al. Coagulation assessment by rotation thrombelastometry in normal pregnancy. Thromb Haemost. 2009;101:755–61.
JPAC Transfusion Handbook (Joint United Kingdom [UK] Blood Transfusion and Tissue Transplantation Services Professional Advisory Committee). 2014. Available at: https://www.transfusionguidelines.org/transfusion-handbook/7-effective-transfusion-in-surgery-and-critical-care/7-3-transfusion-management-of-major-haemorrhage (accessed April 2019).
Karkouti K, McCluskey S, Syed S, Pazaratz C, Poonawala H, Crowther M. The influence of perioperative coagulation status on postoperative blood loss in complex cardiac surgery: A prospective observational study. Anesth Analg. 2010;110:1533–40.
Kassebaum NJ, Bertozzi-Villa A, Coggesshall MS. Global, regional, and national levels and causes of maternal mortality during 1990–2013; a systematic analysis for the Global Burden of Disease Study. Lancet. 2014;384:980–1004.
Kindo M, Hoang Minh T, Gerelli S, Perrier S, Meyer N, Schaeffer M et al. Plasma fibrinogen level on admission to the intensive care unit is a powerful predictor of postoperative bleeding after cardiac surgery with cardiopulmonary bypass. Thromb Res. 2014;134:360–8.
Kohler HP, Grant PJ. Plasminogen-activator inhibitor type 1 and coronary artery disease. N Engl J Med. 2000;15:1792–801.
Korte W, Feldges A. Increased prothrombin activation in a patient with congenital afibrinogenemia is reversible by fibrinogen substitution. Clin Investig. 1994;72:396–98.
Kozek-Langenecker SA, Ahmed AB, Afshari A, Albaladejo P, Aldecoa C, Barauskas G et al. Management of severe perioperative bleeding: guidelines from the European Society of Anaesthesiology: First update 2016. Eur J Anaesthesiol. 2017;34:332–95.
Lak M, Keihani M, Elahi F, Peyvandi F, Mannucci PM. Bleeding and thrombosis in 55 patients with inherited afibrinogenaemia. Br J Haematol. 1999;107:204–06.
Levy JH, Tanaka KA, Steiner ME. Evaluation and management of bleeding during cardiac surgery. Curr Hematol Rep. 2005;4:368–72.
Liu J, Sun L, Wang J, Ji G. The relationship between fibrinogen and in-hospital mortality in patients with type A acute aortic dissection. Am J Emerg Med. 2018;36:741–4.
Laurens N, Koolwijk P, de Maat MP. Fibrin structure and wound healing. J Thromb Haemost. 2006;4:932–9.
Maegele M, Gu ZT, Huang QB, Yang H. Updated concepts on the pathophysiology and the clinical management of trauma hemorrhage and coagulopathy. Chin J Traumatol. 2017;20:125–32.
Makar M, Taylor J, Zhao M, Farrohi A, Trimming M, D’Attellis N et al. Perioperative coagulopathy, bleeding, and hemostasis during cardiac surgery: a comprehensive review. ICU Director. 2010;1:17–27.
Mosesson MW. Antithrombin I. Inhibition of thrombin generation in plasma by fibrin formation. Thromb Haemost. 2003;89:9–12.
Moulton MJ, Creswell LL, Mackey ME, Cox, JL, Rosenbloom M. Reexploration for bleeding is a risk factor for adverse outcomes after cardiac operations. J Thorac Cardiovasc Surg. 1996;111:1037–46.
Nacca CR, Shah KN, Truntzer JN, Rubin LE. Total knee arthroplasty in a patient with hypofibrinogenemia. Arthroplasty. 2016;2:177–82.
Neerman-Arbez, M., Casini, A. Clinical Consequences and Molecular Bases of Low Fibrinogen Levels. International Journal of Molecular Sciences. 2018;19:1.
Niepraschk-von Dollen K, Bamberg C, Henkelmann A, Mickley L, Kaufner L, Henrich W et al. Predelivery maternal fibrinogen as a predictor of blood loss after vaginal delivery. Arch Gynecol Obstet. 2016;294:745–51.
O'Riordan MN, Higgins JR. Haemostasis in normal and abnormal pregnancy. Best Pract Res Clin Obstet Gynaecol. 2003;17:385–96.
Oruc N, Tokat Y, Killi R, Tombuloglu M, Ilter T. Budd-Chiari syndrome in an afibrinogenemic patient: a paradoxical complication. Dig Dis Sci. 2006;51:378–80.
Palmer JD, Francis DA, Roath OS, Francis JL, Iannotti F. Hyperfibrinolysis during intracranial surgery: effect of high dose aprotinin. J Neurol Neurosurg Psychiatry. 1995;58:104–6.
Peyvandi F, DiMichele D, Bolton-Maggs PH, Lee CA, Tripodi A, Srivastava A. Classification of rare bleeding disorders (RBDs) based on the association between coagulant factor activity and clinical bleeding severity. J Thromb Haemost. 2012;10:1938–43.
Roberts I, Shakur H, Fawole B, Kuti M, Olayemi O, Bello A et al. Haematological and fibrinolytic status of Nigerian women with post-partum haemorrhage. BMC pregnancy and childbirth. 2018;18:143.
Sahni A, Odrljin T, Francis CW. Binding of basic fibroblast growth factor to fibrinogen and fibrin. J Biol Chem. 1998;273:7554–59.
Saner FH, Kirchner C. Monitoring and Treatment of Coagulation Disorders in End-Stage Liver Disease. Visc Med. 2016;32:241–48.
Scrutinio D, Giannuzzi P. Comorbidity in patients undergoing coronary artery bypass graft surgery: impact on outcome and implications for cardiac rehabilitation. European Journal of Preventative Cardiology. 2008;15:379–85.
Simon L, Santi TM, Sacquin P, Hamza J. Pre-anaesthetic assessment of coagulation abnormalities in obstetric patients: usefulness, timing and clinical implications. Br J Anaesth. 1997;78:678–83.
Tennent GA, Brennan SO, Stangou AJ, O’Grady J, Hawkins PN, Pepys MB. Human plasma fibrinogen is synthesised in the liver. Blood. 2007;109:1971–4.
Teufelsbauer H, Proidl S, Havel M, Vukovich T. Early activation of hemostasis during cardiopulmonary bypass: evidence for thrombin mediated hyperfibrinolysis. Thromb Haemost. 1992;68:250–2.
Theusinger OM, Wanner GA, Emmert MY, Billeter A, Eismon J, Seifert B et al. Hyperfibrinolysis diagnosed by rotational thromboelastometry (ROTEM) is associated with higher mortality in patients with severe trauma. Anesth Analg. 2011;113:1003–12.
Tziomalos K, Vakalopoulou S, Perifanis V, Garipidou V. Treatment of congenital fibrinogen deficiency: overview and recent findings. Vasc Health Risk Manag. 2009;5:843–8.
Venugopal A. Disseminated intravascular coagulation. Indian J Anaesth. 2014;58:603–8.
Vivacqua A, Koch CG, Yousuf AM, Nowicki ER, Houghtaling PL, Blackstone EH et al. Morbidity of bleeding after cardiac surgery: is it blood transfusion, reoperation for bleeding, or both? Ann Thorac Surg. 2011;91:1780–90.
Walden K, Jeppsson A, Nasic S, Backlund E, Karlsson M. Low Preoperative Fibrinogen Plasma Concentration Is Associated With Excessive Bleeding After Cardiac Operations. Ann Thorac Surg. 2014;97:1199–206.
Developed by EPG Health for Medthority in collaboration with CSL Behring, with content provided by CSL Behring.
Not intended for Healthcare Professionals outside Europe.